
Adipic acid is a linear dicarboxylic acid, presenting as a white crystalline compound under standard temperature and pressure conditions. It holds a significant position within the chemical industry, consistently ranking among the top 10 chemicals in terms of annual volume consumption. Globally, approximately 2.5 million tons of adipic acid are produced each year. The primary application of adipic acid lies in the synthesis of 6,6 nylon, with additional uses encompassing resins, plasticizers, lubricants, polyurethanes, and food additives.
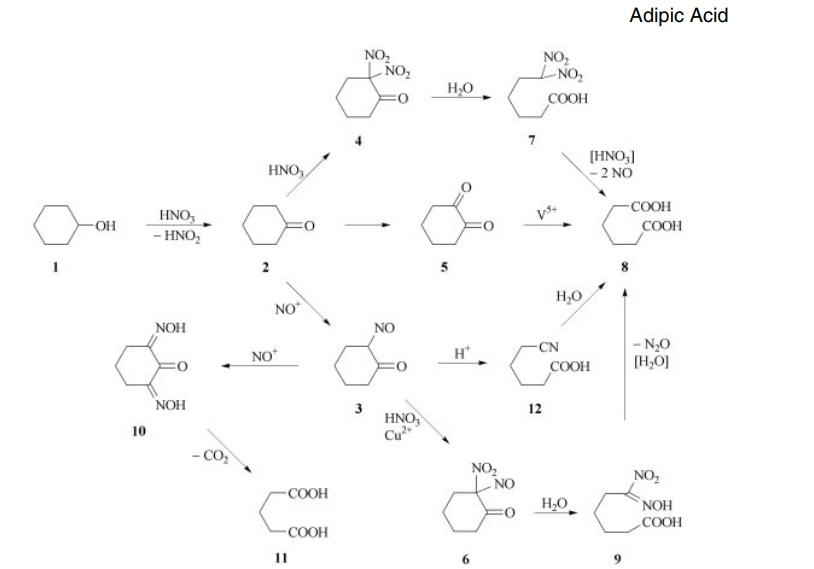
Various methods can be employed in the production of adipic acid, with the traditional and predominant approach being the two-step oxidation of cyclohexane, as illustrated in Figure. The initial step involves the oxidation of cyclohexane to cyclohexanone and cyclohexanol, utilizing oxygen or air, and catalyzed by cobalt or manganese at around 150°C. Subsequently, the second oxidation, employing nitric acid and air with copper or vanadium catalysts, opens the ring structure to form adipic acid and nitrous oxide. Alternative feedstocks, such as benzene and phenol, may be utilized in the synthesis of adipic acid. Measures to control nitrous oxide emissions, a greenhouse gas, have been implemented through pollution abatement technology in recent years.

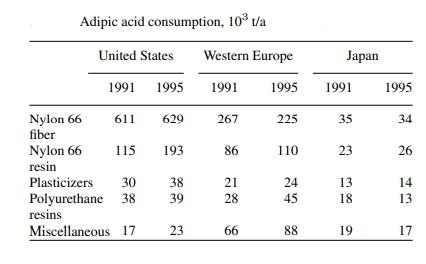
The predominant application of adipic acid, constituting approximately 90% of its usage, is in the production of nylon. Nylon, the pioneer truly synthetic fiber, marked a significant advancement in materials for clothing and other purposes. Historically, fibers such as wool, leather, cotton, flax, and silk were limited to providing fabric. The advent of synthetic fibers began in the late 19th century, initially involving chemical formulations utilizing cellulose. Rayon, the first synthetic fiber produced commercially, emerged through the nitration of cellulose, with various production methods evolving over time.
By the 1920s, several companies were engaged in producing cellulose-based synthetic materials, setting the stage for truly synthetic materials. DuPont, a chemical company with a diversified portfolio, entered the synthetic fiber arena in the 1920s, investing significantly in research on synthetics. In 1935, DuPont introduced Fiber 66, the first nylon, produced by reacting adipic acid and hexamethylenediamine. Nylon, a term coined in 1938, represented a group of synthetically produced polyamides. Despite attempts to keep the name secret until the 1939 World's Fair, DuPont eventually disclosed it due to leaks and patent preparations, promoting the material generically as nylon.
Nylon 66, originally named Fiber 66, is synthesized through the combination of adipic acid and hexamethylene-diamine under specific conditions:

Between 1936 and 1939, DuPont dedicated efforts to developing production methods and marketing strategies for nylon. Unfortunately, the inventor of nylon, Wallace Carothers, did not live to witness the success of his creation, as he took his own life in April 1937. Nylon initially gained popularity as a substitute for silk in women's hosiery. Its debut as a significant technological advancement occurred at the 1939 World's Fair in New York City, although it had been utilized in toothbrush bristles over a year prior. By 1941, nylon found applications in neckties, toothbrushes, thread, and certain garments. During World War II, the U.S. government exclusively requisitioned nylon production for wartime purposes, replacing silk in military items like parachutes, tents, rope, and tires. Post-war, nylon's civilian applications, including nylon stockings, resumed. Apart from its role as a hosiery fabric, nylon extended its utility to upholstery, carpeting, nets, and clothing.
Adipic acid can alternatively polymerize with alcohols like ethylene glycol to yield polyesters, which can then combine with isocyanates to form polyurethanes. Smaller esters of adipic acid, generated with alcohols in the C-8 to C-10 range, are termed adipates. These adipates function as softeners in plastics, such as polyvinyl chloride, and as synthetic grease base oils. Adipic acid also finds application in the food industry, where food-grade adipic acid is either synthetically produced or extracted from beet juice as a natural source. Its uses in the food industry include serving as a gelling agent, an acidulant to impart tartness, and a preservative.
Richard L. Myers (2009). The 100 Most Important Chemical Compounds: A Reference Guide. Greenwood Publishing Group. October 1, 2009. https://doi.org/10.1021/ed086p1182
 |
 |
 |