It is common for a sign to be placed outside street pharmacies, with the words “Viagra is in stock” prominently displayed.
What exactly is "Viagra"? Why does it occupy such a prominent advertising spot in pharmacies?
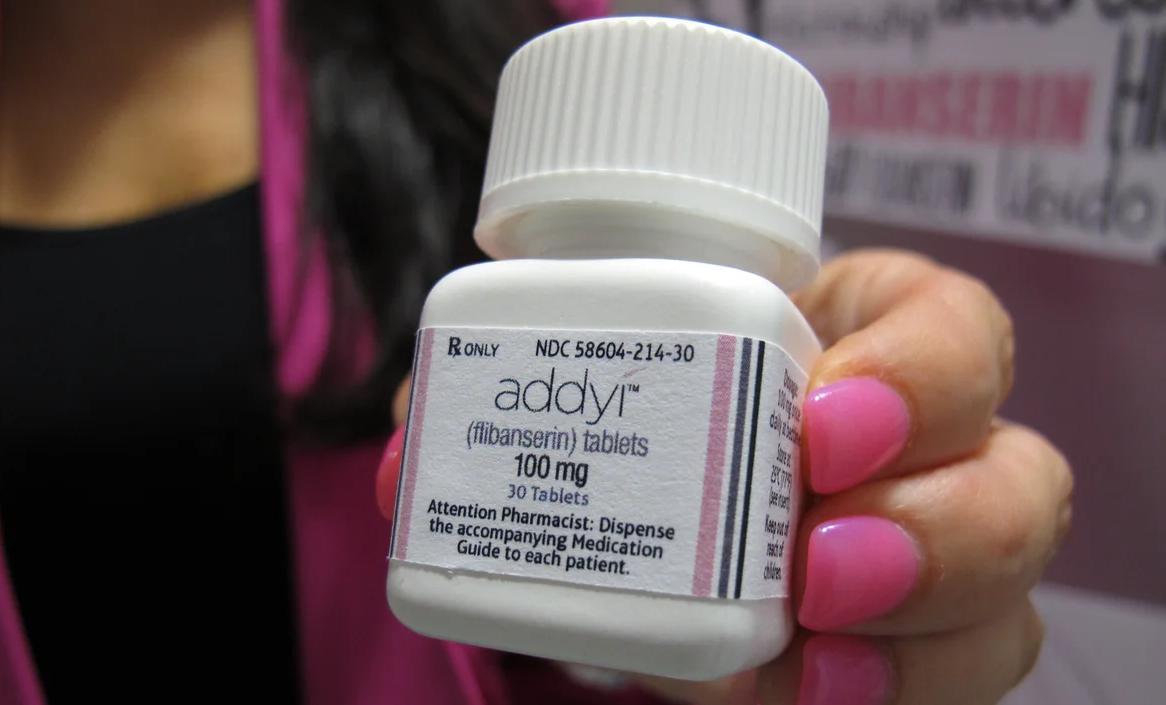
In fact, "Viagra" is what people commonly refer to as “the little blue pill.” When it was released in 1998, it brought a multi-billion-dollar boom to the male sexual health market. However, while countless male patients benefited from it, many women suffering from sexual dysfunction also hoped that something like "Viagra for women" could save their lives. Against this background, a pink pill—Addyi—was born.
Unfortunately, this drug did not spark a revolution in the sexual health field like "Viagra" did. On the contrary, since its inception, it has been embroiled in decades of heated debates, and its development trajectory has reflected the magical spectrum created when scientific rationality, capital ambition, and social movements collide.
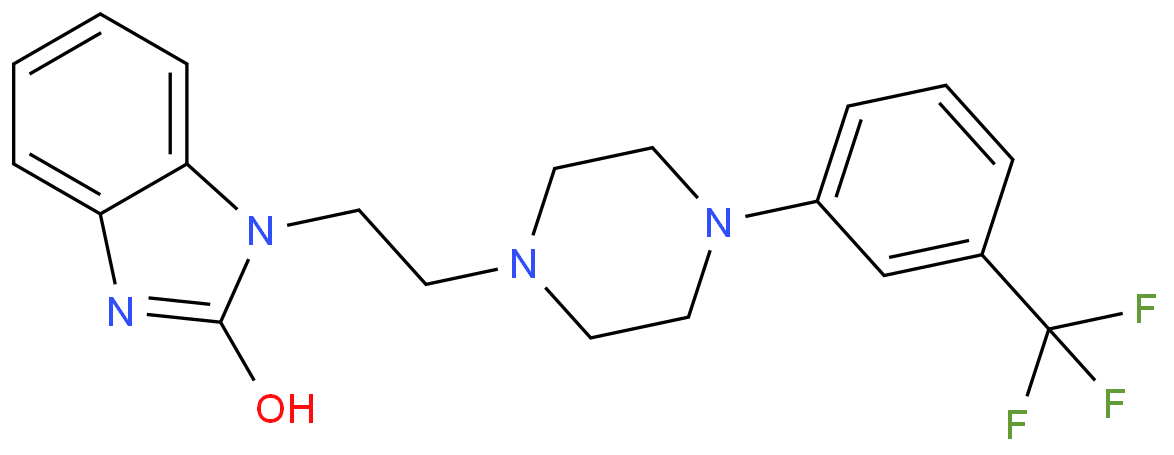
Addyi’s generic name is flibanserin, and it was initially developed by the German pharmaceutical giant Boehringer Ingelheim in the 1990s as an antidepressant. The original design was based on the serotonin (5-HT) receptor modulation mechanism, aiming to treat depression by adjusting the balance of neurotransmitters in the brain.
However, subsequent clinical trials showed that the drug was ineffective against depression, to the point of being essentially useless. This was discouraging for the researchers, and Boehringer Ingelheim considered halting further development. During this difficult time, researchers noticed an interesting phenomenon: many female depression patients reported an increase in sexual desire during the course of taking the drug.
At that time, Female Sexual Interest/Arousal Disorder (FSIAD) was gaining attention in the medical field. FSIAD, which falls under the category of female sexual dysfunction, is a mental-genital disorder that can affect women of all ages. Epidemiological surveys showed that the prevalence of FSIAD in premenopausal women worldwide was approximately 10%. Given the large number of patients and the limited effectiveness of traditional drug treatments, the unexpected discovery of flibanserin was a pleasant surprise for the research team, leading them to reconsider the drug’s potential application. This scenario seems quite similar to the case of "Viagra."
Further studies revealed that flibanserin’s mechanism of action was not the traditional single pathway of antidepressants, but rather a unique "dual receptor modulation" characteristic, meaning it both agonizes the 5-HT1A receptor and antagonizes the 5-HT2A receptor (blocking this receptor reduces its negative regulation on sexual desire). Additionally, it can indirectly promote the release of dopamine and norepinephrine, neurotransmitters that help increase sexual arousal and improve attention.
With this new research direction, Boehringer Ingelheim invested heavily in human trials of flibanserin over the next few years, focusing on women with FSIAD.
In 2009, Boehringer Ingelheim submitted its Phase III clinical trial data to the FDA with high hopes for approval. However, after reviewing the application, the FDA decisively rejected it, citing that while self-reported data showed an increase in the average number of sexual satisfaction events per month (from 2.8 to 4.5), the placebo group also showed a similar improvement (from 2.7 to 3.7). Therefore, after accounting for the placebo effect, the drug’s efficacy was only a modest increase of 0.7 events per month. Furthermore, the FDA’s preferred data from electrode-recording instruments showed no significant difference between flibanserin and the placebo.
After this setback, Boehringer Ingelheim was thoroughly disheartened, and in 2011, they sold flibanserin to another company, Sprout Pharmaceuticals, altering the drug’s fate.
Upon acquiring flibanserin, Sprout immediately assessed the FDA’s objections. Perhaps after intense internal discussions or consulting with "experts," they made an unprecedented decision: first, they would expand the sample size for further trials, hoping to bolster the argument with more self-reported data; second, they would cancel the electrode-recording data to prevent the FDA from using it against them. This tactic seemed akin to finding loopholes.
In 2013, new clinical trial data emerged. As Sprout had anticipated, self-reports indicated that, after 24 weeks of taking flibanserin, patients had an average increase of 0.5 sexual satisfaction events per month, consistent with previous results. However, its side effects were startling, including low blood pressure, drowsiness, dizziness, and nausea. Despite these serious side effects, Sprout pressed ahead with submitting the application to the FDA. But the FDA was not easily swayed. Sprout’s clever pharmacological endpoints failed to convince experts in the field, and the severe side effects could not be ignored. In the end, the FDA rejected the new drug application for flibanserin once again.
Despite being rejected by the FDA twice, Sprout remained remarkably persistent and unwilling to let its efforts go to waste. After another round of internal discussions or consultations with "experts," they developed a strategy that would secure their position: they decided to play the gender equality card.
Sprout noted that in the past few decades, the FDA had approved 26 drugs for treating male sexual dysfunction, but had never approved a drug for female sexual dysfunction. Was this fair? Was this not blatant discrimination against women? Regardless of the facts, such a statement was politically correct and impossible to dismiss. With this breakthrough, Sprout began an aggressive media campaign.
In 2013, Sprout funded the creation of a gender equality movement called "Even the Score." The organization not only widely publicized the alleged gender bias of the FDA in new drug evaluations on social media but also wrote letters to members of Congress demanding an investigation into the FDA’s actions. This politically charged bombshell caught the FDA off guard, forcing it to clarify that the 26 drugs approved for male sexual dysfunction were, in fact, just different formulations of the same drug, and the FDA had also never approved any drug for male sexual desire disorders. Thus, no discrimination existed.
Riding this wave of public opinion, Sprout submitted its new application for flibanserin again. In June 2015, after a long hearing and debate, the FDA’s advisory committee finally voted 18 to 6 in favor of the drug, concluding that its benefits outweighed its risks. In August of the same year, the FDA officially approved flibanserin (Addyi) for the treatment of hypoactive sexual desire disorder in premenopausal women, with the requirement that it be labeled with the most severe "black box warning" regarding the drug’s serious side effects.
The day after Addyi’s approval, Even the Score posted a celebratory video on social media and thanked the FDA. Perhaps having accomplished its mission, the account disappeared from the internet after posting its final tweet in January 2016, and the website became inaccessible.
Perhaps drawing inspiration from "Viagra" and imagining an unlimited future for flibanserin, Valeant Pharmaceuticals announced two days after its approval that it would acquire Sprout for one billion dollars. However, the harsh reality soon became evident. Data showed that Addyi’s sales in the first four months after its launch amounted to only $9 million, while "Viagra" earned an astounding $320 million during the same period. It seemed that Valeant’s dream of replicating Viagra’s sales miracle was nothing more than a fantasy.
Not only that, the medical community’s criticism of flibanserin grew louder. In 2015, the Journal of the American Medical Association published an article pointing out that flibanserin’s pharmacological data was highly flawed and incomplete. In 2016, the Journal of Internal Medicine criticized the drug’s minimal efficacy and significant toxicity. In 2018, the Journal of Industrial Psychiatry questioned whether hypoactive sexual desire disorder even existed...
If the "disease" didn’t exist, how could the "drug" be justified? The troubled history of flibanserin, though resembling a farce, reveals the complex ecology of drugs moving from the laboratory to the market. It inevitably sparks deep reflection on scientific ethics, gender equality, and the power of capital.
[4]. Evaluation of Flibanserin: Science and Advocacy at the FDA.