On April 25th, Roche Group announced that the New England Journal of Medicine (NEJM) published a detailed analysis of the Phase III CENTERSTONE clinical trial for Xofluza? (Baloxavir Marboxil). The study confirmed that a single oral dose of Xofluza? significantly reduced the likelihood of untreated household members contracting the virus by 32% (primary endpoint). In terms of the key secondary endpoint (influenza virus transmission rate in symptomatic individuals), Xofluza? did not reach statistical significance but showed a clinically meaningful trend of reduction. The drug was well tolerated, and no new safety signals were observed.
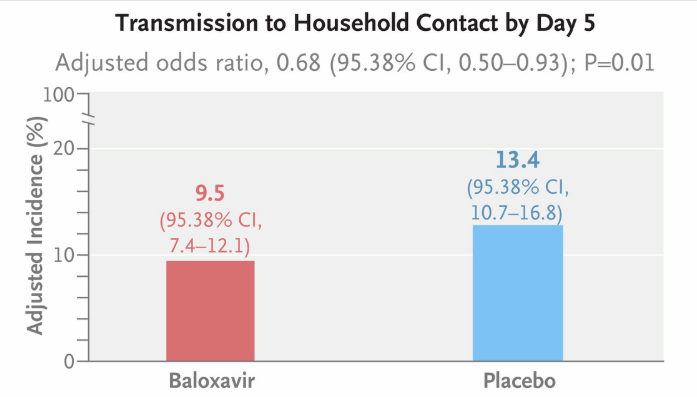
During the 2019-2024 influenza seasons, 1,457 index patients and 2,681 household contacts were included. Among them, 726 index patients were assigned to the baloxavir group, and 731 were assigned to the placebo group. The results showed that, on Day 5, the laboratory-confirmed influenza transmission rate (primary efficacy endpoint) in the baloxavir group was significantly lower than that of the placebo group (adjusted incidence rate 9.5% vs. 13.4%; adjusted odds ratio 0.68; 95.38% confidence interval [CI] 0.50-0.93; P=0.01), with a 29% adjusted relative risk reduction (95.38% CI 12-45). Additionally, the influenza virus transmission rate for symptomatic individuals on Day 5 was 5.8% in the baloxavir group and 7.6% in the placebo group, with no statistical difference (secondary endpoint not achieved). As for safety, 7.2% (95% CI 4.1-11.6) of the index patients in the baloxavir group had detectable resistant viruses during follow-up, while no resistant viruses were detected in the household contacts, and no new safety signals were identified.

Roche stated that these clinical trial results have been submitted for review to regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Influenza poses a significant burden on public health and the economy, especially for high-risk populations with influenza-related complications. Globally, seasonal influenza causes about 1 billion infections annually, millions of hospitalizations, and up to 650,000 deaths. Approximately one-third of influenza virus transmission occurs within households. Up to 75% of working adults may experience an average of two days of absenteeism due to illness in themselves or their family members, yet most report continuing to work even with symptoms. In the event of an influenza pandemic, it could have a severe impact on the overall functioning of healthcare systems. With the co-circulation and increased disease burden of multiple respiratory viruses (including COVID-19) during the winter and non-seasonal periods, having effective treatments and solutions to prevent influenza transmission is more important than ever.
Levi Garraway, MD, Chief Medical Officer and Head of Global Product Development at Roche, stated: "This study is the first to demonstrate that an antiviral drug can reduce the risk of influenza virus transmission within households. These results may have far-reaching implications for public health. The research highlights the urgent need for solutions that can alleviate the burden of influenza on society."
[1]. Timothy M. Uyeki,Vivien G. Dugan,Demetre C. Daskalakis,Baloxavir Treatment to Reduce Influenza Virus Transmission, New England Journal of Medicine, 392, 16, (1652-1654), (2025). /doi/full/10.1056/NEJMe2503242