Guanidine nitrate, as a significant chemical substance, has sparked widespread interest and research. Its unique properties and extensive applications have made it a focal point in the chemical industry.
Chemically known as diamino guanidine nitrate, with the molecular formula CH6N4O3 or CH5N3·HNO3, and a molecular weight of 122.08, guanidine nitrate exists in various forms such as crude guanidine nitrate, refined guanidine nitrate, specialty ultrafine guanidine nitrate, and reagent-grade. It is a novel fine chemical product with broad applications. In the production processes of industries like dyes and pesticides, it serves as a crucial intermediate. Moreover, it is a new material for automotive safety airbags and finds extensive use in disinfectants, explosives, paints, and other fields. The structural formula of guanidine nitrate is as follows:
Guanidine nitrate appears as white crystalline powder or granules. It is an organic strong base and a powerful oxidizing agent with moderate toxicity, having a melting point of 217°C. What is the solubility of guanidine nitrate in water? The solubility of guanidine nitrate in water increases with temperature. For instance, at 20°C, 12.5 g of guanidine nitrate completely dissolves in 100 g of water, while at 80°C, 99 g dissolves in 100 g of water. Its solubility in ethanol follows a similar trend. It must adhere to a quality standard content of 98%, maintaining a neutral pH. Due to its oxidizing properties, it may explode when heated, exposed to an open flame, or subjected to friction, vibration, or impact. Mixtures with nitro compounds and chlorates are sensitive to shock and friction, also leading to explosions. Upon high-temperature decomposition, toxic combustion products like nitrogen oxides are produced.
Guanidine nitrate finds extensive applications not only in the chemical industry but also in medicine, agriculture, and high-tech industries. It serves as an excellent gas-generating agent and a key intermediate for various compounds, acting as an excellent nitrating agent, additive, and precipitant. Additionally, it is utilized as a new material for automotive safety airbags and in medicine for synthesizing drugs like loflupren hydrochloride. Guanidine nitrate is also widely used in rocket propellants, paint industry, photographic materials, and disinfectants. Furthermore, it serves as a versatile chemical raw material, acting as a crucial intermediate for producing drugs like sulfathiazole and sulfadimidine. In the pesticide industry, it not only serves as a raw material for insecticide pyrimethanil but also as an intermediate for various sulfonylurea herbicides and in manufacturing composite explosives like cast, screw-loaded, and plastic-bonded explosives. It can act as an effective additive in dye-sensitized solar cell electrolytes and a good precipitant for separating tungsten and molybdenum in the form of tungsten. It can synthesize a new type of guanidine-based bisazo fluorescent chemical sensor, L. With the rapid development of the pharmaceutical and pesticide industries, the domestic demand for guanidine nitrate is increasing, with promising prospects not only for domestic industrial production but also for export.
Different countries employ various methods for guanidine nitrate production. The United States and Japan mainly use the dicyandiamide method, while Germany employs the direct method, producing guanidine nitrate directly from aminoguanidine. The dicyandiamide method is the main method in traditional processes, characterized by high yield. The urea method is developed from the dicyandiamide method, using urea instead of dicyandiamide for preparation, with easier access to raw materials and lower costs but with adverse reactions affecting the preparation. Additionally, there are methods such as continuous flow synthesis of nitroguanidine using microchannels and the cyanamide-calcium method. Currently, mature guanidine nitrate production processes in China include the dicyandiamide method, urea method, and BAF method. Compared to the urea method and BAF method, the dicyandiamide method has the easiest reaction conditions, no by-products, simple post-treatment, and lower costs. Therefore, the stable dicyandiamide method was selected as the research scheme for this study.
Guanidine nitrate, a compound derived from guanidine and nitric acid, has various industrial uses. One of its primary applications is in propellants and explosive formulations. Due to its high nitrogen content, guanidine nitrate is typically added to solid rocket propellants as a fuel and aids in sustaining combustion. Additionally, it is utilized in certain pharmaceutical formulations, particularly as a precursor for drugs and drug intermediates. Its versatility extends to its role as an organic chemical reagent, facilitating the synthesis of various organic compounds.
While guanidine nitrate offers many industrial advantages, its handling requires strict adherence to safety protocols. This compound carries inherent risks, including toxicity and reactivity. Direct contact with guanidine nitrate can cause irritation to the skin, eyes, and respiratory system. Additionally, it may decompose under certain conditions, leading to the release of toxic gases or even the risk of explosion.
Prudent handling of guanidine nitrate necessitates comprehensive risk mitigation measures. The compound must be stored in a cool, dry place away from heat sources or open flames. Proper ventilation in storage areas helps prevent the accumulation of potentially hazardous vapors. During handling and transportation, containers should be securely sealed and labeled to ensure clear identification and minimize the risk of accidental exposure. Personnel handling guanidine nitrate must wear appropriate personal protective equipment such as gloves, goggles, and respirators to minimize contact risks, adhering to the guanidine nitrate SDS.
In this article, we delved into the properties and applications of guanidine nitrate. As a versatile compound, it plays an irreplaceable role in military, medical, and industrial fields. With the continuous advancement of science and technology, it is believed that the application scope of guanidine nitrate will continue to expand, making new contributions to human development and progress.
[1] Yu Xiaohong. Research on process for improving the yield of guanidine nitrate [J]. Contemporary Chemical Research, 2024, (05): 176-178. DOI:10.20087/j.cnki.1672-8114.2024.05.056.
[2] Hu Donglin. Analysis of production method and application of guanidine nitrate [J]. Shanxi Chemical Industry, 2022, 42 (01): 40-41+44. DOI:10.16525/j.cnki.cn14-1109/tq.2022.01.014.
[3] Wang Xuezhi. Study on thermal runaway mechanism of guanidine nitrate [D]. China University of Petroleum (East China), 2017.
[4] https://pubchem.ncbi.nlm.nih.gov/compound/10481
 |
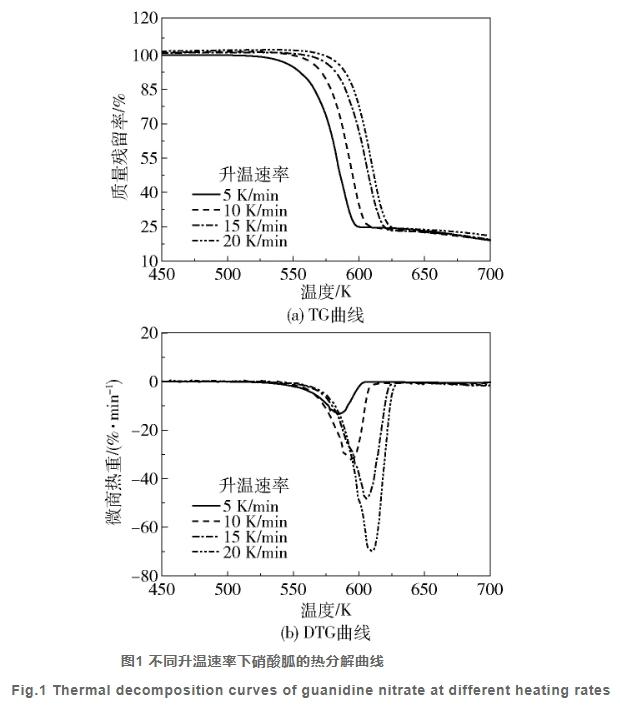 |
 |