Delving into the complexity of the hexamethylenetetramine mechanism provides a deeper understanding of its functionality across various applications and industries.
Hexamethylenetetramine (HMT), also known as methenamine or urotropin, is a heterocyclic organic compound with the molecular formula (CH2)6N4. This white crystalline compound is highly soluble in water and polar organic solvents. It features a cage-like structure analogous to adamantane, with its four carbon atoms replaced by nitrogen atoms linked via methylene bridges (CH2 groups).
Hexamethylenetetramine has the chemical formula (CH2)6N4, forming a tetrahedral cage structure with nitrogen atoms replacing carbon atoms found in adamantane.
- Molecular mass: 140.186 g/mol
- Physical appearance: White crystalline solid
- Odor: Faint, sometimes described as fishy or ammoniacal
- Solubility: Soluble in water, chloroform, methanol, ethanol, acetone, benzene, xylene, ether
- Melting point: Sublimes at 280 ℃ (536 °F)
- Density: 1.33 g/cm3 (at 20 ℃)
- Acidity (pKa): 4.89
Hexamethylenetetramine behaves akin to amine bases, undergoing protonation and N-alkylation (e.g., alkylating with allyl chloride to form quaternary ammonium salts).
While generally safe when handled properly, hexamethylenetetramine can irritate the skin and eyes upon contact. Inhalation of dust or fumes should be avoided. Observing safety guidelines is crucial when working with this compound.
(1) Acidic Environment: When hexamethylenetetramine enters the acidic environment of urine, it slowly releases formaldehyde, a compound with preservative properties.
(2) Bactericidal Action: Formaldehyde disrupts bacterial protein synthesis, inhibiting bacterial growth and causing infection. While hexamethylenetetramine itself is not directly antimicrobial, in the acidic environment (pH < 6) of urine, it releases formaldehyde slowly. Formaldehyde is an effective disinfectant, denaturing proteins and nucleic acids within bacteria, ultimately killing them. Hence, hexamethylenetetramine is effective against urinary tract infections caused by bacteria sensitive to acidic urine.
It's important to note that hexamethylenetetramine is not a first-line treatment for urinary tract infections and should only be used under medical supervision.
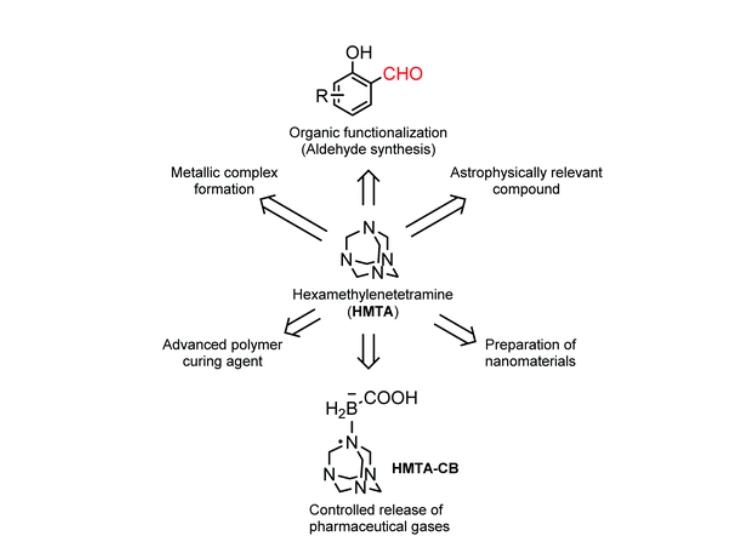
(1) Duff Reaction: Utilizing hexamethylenetetramine to introduce a formyl group (CHO) into aromatic compounds (arenes), such as the synthesis of cinnamaldehyde.

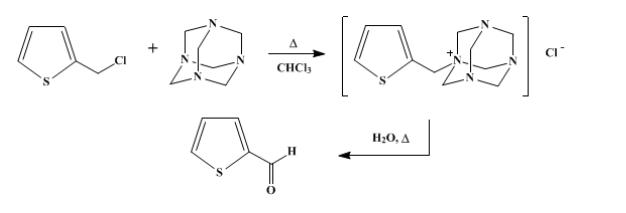
(2) Sommelet Reaction: Hexamethylenetetramine facilitates the conversion of benzyl halides to aldehydes, useful intermediates in organic synthesis, such as the synthesis of 2-thiophenealdehyde.
(3) Delepine Reaction: This reaction utilizes hexamethylenetetramine to synthesize amines from alkyl halides, for example, the synthesis of 2-bromoallylamine.
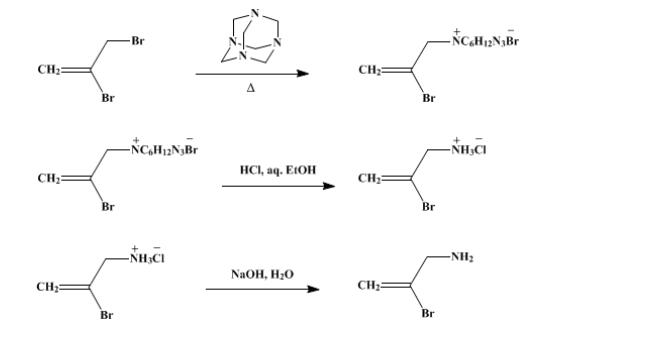
These reactions highlight the practicality of hexamethylenetetramine as a building block or catalyst in the construction of complex organic molecules.
Hexamethylenetetramine is a multifunctional compound with wide-ranging applications across different industries. Its unique chemical structure and properties enable it to function in various ways, making it a valuable material.
Discovered by Aleksandr Butlerov in 1859, hexamethylenetetramine is industrially synthesized by the reaction of formaldehyde and ammonia, involving a series of condensation reactions leading to the formation of HMT.
In conclusion, while significant progress has been made in understanding the hexamethylenetetramine mechanism, many questions remain unresolved. Its widespread applications underscore the importance of further research into its mechanisms, particularly in biomedical and environmental fields, to guide its safe and effective use.
[1] Allen CH, Leubne GW (1951). "Syringic Aldehyde". Organic Syntheses. 31: 92. doi:10.15227/orgsyn.031.0092.
[2]Wiberg KB (1963). "2-Thiophenaldehyde". Organic Syntheses. doi:10.15227/orgsyn.000.0000; Collected Volumes, vol. 3, p. 811.
[3]Bottini AT, Dev V, Klinck J (1963). "2-Bromoallylamine". Organic Syntheses. 43: 6. doi:10.15227/orgsyn.043.0006.
[4]https://en.wikipedia.org/wiki/Hexamethylenetetramine
[5]Eller K, Henkes E, Rossbacher R, H?ke H (2000). "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH. doi:10.1002/14356007.a02_001. ISBN 9783527306732.
[6]https://www.organic-chemistry.org/namedreactions/delepine-reaction.shtm
[7]https://pubchem.ncbi.nlm.nih.gov/compound/4101
 |
 |
 |