Polyacrylonitrile is an important synthetic polymer with wide-ranging applications including textiles, synthetic fibers, and membrane materials. Its unique structure and properties make it a material of significant interest in both industrial and research fields. However, understanding the structure of polyacrylonitrile may not be straightforward for beginners. Hence, this article aims to provide you with a comprehensive guide to help you understand the structural characteristics and properties of polyacrylonitrile. Through this introduction, you will gain a better understanding of this important material, aiding in your learning and research endeavors.
Polyacrylonitrile, commonly abbreviated as PAN, is a versatile synthetic polymer known for its strength and durability. PAN fibers are lightweight, abrasion-resistant, chemically resistant, and can withstand high temperatures. They also exhibit good thermal stability, maintaining their shape well at elevated temperatures. This unique combination of properties makes PAN valuable in many industries.
In the textile industry, PAN is used to produce sportswear, carpets, and high-performance fabrics for industrial applications. Its strength and durability make it an ideal material for tire cords in the automotive industry. One of PAN's most important applications is as a precursor material for carbon fibers, which are widely used in aerospace and high-performance engineering.
Polyacrylonitrile (PAN) is a semi-crystalline organic polymer with the molecular formula (C3H3N)n and features cyano (CN) functional groups attached to the polyethylene backbone as structural units. The structure of polyacrylonitrile is depicted in the following diagram. The cyano groups act as hydrogen bond acceptors due to the lone pair of electrons on the nitrogen atom and exhibit a significant dipole moment between the electron-deficient carbon atom and the electron-rich nitrogen atom, allowing them to engage in relatively strong attractive interactions. Indeed, strong intermolecular interactions result in high strength and tolerance to various organic solvents.
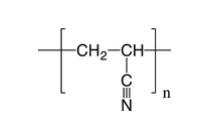
The polyacrylonitrile formula of PAN is (C3H3N)n. This formula represents the repeating units within the polymer chain. "n" indicates that these repeating units can connect multiple times to form a long chain molecule. Polyacrylonitrile (PAN) being a polymer means it is composed of many smaller repeating units.
The molecular formula provides the basic building blocks of PAN. It informs us of the exact number of atoms of each element (carbon, hydrogen, and nitrogen) in each repeating unit, which is crucial for understanding possible chemical reactions of PAN and predicting its properties.
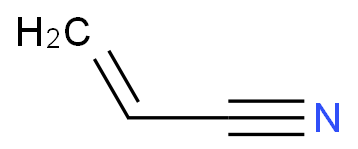
PAN is composed of acrylonitrile monomers linked together. The chemical formula of acrylonitrile itself is CH2CHCN.
The repeating units in the PAN chain consist of a three-carbon backbone (CH2-CH) and a cyano group (CN) attached to the middle carbon.
The presence of the cyano group (CN) is crucial for PAN's properties. This group is polar, meaning its electron distribution is uneven, giving PAN some chemical resistance and affinity to certain solvents. The alternating single and double bonds in the chain structure enhance PAN's strength and rigidity.
The molar mass of polyacrylonitrile is 53.0626 ± 0.0028 g/mol (C 67.91%, H 5.7%, N 26.4%). The polyacrylonitrile density is 1.184 g/cm3, with a melting point of 300 ℃ (572 °F; 573 K). Although it is a thermoplastic, polyacrylonitrile does not melt under normal conditions; it degrades before melting. It melts above 50 ℃ if the heating rate is 300 degrees per minute or higher. The glass transition temperature is approximately 95℃, and the melting temperature is 322℃. PAN is soluble in polar solvents such as dimethylformamide, dimethylacetamide, ethylene carbonate, and acrylonitrile carbonate, as well as aqueous solutions of sodium thiocyanate, zinc chloride, or nitric acid. Solubility parameters: 26.09 MPa1/2 (25 ℃) ranging from 25.6 to 31.5 J1/2 cm?3/2. Dielectric constants: 5.5 (1 kHz, 25 ℃), 4.2 (1 MHz, 25 ℃). It can behave as both branched and linear polymers.
Polyacrylonitrile is produced from acrylonitrile (CH2=CHCN) through the reaction of acrylonitrile (CH2=CHCH3) with ammonia (NH3) and oxygen in the presence of a catalyst. Acrylonitrile monomers (single-unit molecules) almost always combine with other monomers, suspended as tiny droplets in water, and polymerize into PAN through the action of free-radical initiators.
PAN is a semi-crystalline polymer, meaning it has both ordered and disordered regions in its structure. Below are the arrangements of atoms and bonds:
The following is a simplified representation of the repeating unit structure of PAN: n(CH2-CH(CN)). "n" represents the number of repeating units, which can vary depending on the length of the polymer chain.
As mentioned earlier, PAN is a semi-crystalline polymer. This means its physical structure consists of a combination of two different regions:
The proportion of amorphous and crystalline regions in PAN affects its overall performance. Higher crystallinity makes PAN stronger and stiffer but less flexible. Conversely, a more amorphous structure increases flexibility but lowers strength.
PAN is often processed into fibers for use in textiles and other applications. The morphology (shape and structure) of these fibers plays a crucial role in their properties:
Pure polyacrylonitrile (PAN) consisting only of acrylonitrile monomers is rarely used commercially. Most PAN resins are copolymers, meaning they contain a small portion of other monomers (usually less than 20%) besides acrylonitrile. By combining different comonomers, manufacturers can tailor PAN's properties for various applications. Compared to pure PAN, copolymers can enhance processability, dyeability, chemical resistance, and thermal behavior.
Apart from copolymers, the structure of PAN can be further modified through various techniques:
Various additives are often incorporated during PAN processing to achieve desired properties:
By combining copolymers, modifications, and additives, manufacturers can create a wide range of PAN-based materials with specific functionalities suitable for different applications.
Through this article, we aim to provide readers with a more comprehensive and in-depth understanding of the structure of polyacrylonitrile. As an important synthetic polymer, polyacrylonitrile finds wide applications across various fields and plays a significant role in materials science and engineering. Understanding the structural characteristics, synthesis methods, and properties of polyacrylonitrile helps us better leverage the advantages of this material and develop more high-performance applications. We hope this article helps you better understand and apply polyacrylonitrile, fostering development and innovation in related fields. If you have any further questions or would like to delve deeper into polyacrylonitrile, feel free to continue researching and exploring.
[1] https://en.wikipedia.org/wiki/Polyacrylonitrile
[2] https://www.britannica.com/science/polyacrylonitrile
[3] https://www.pslc.ws/macrog/kidsmac/polyac.htm
[4] https://www.igtpan.com/Ingles/propriedade-poli.asp
[5] https://link.springer.com/referenceworkentry/10.1007/978-3-642-29648-2_249
 |
 |
 |