Xylene solvent, mainly derived from the widespread use in home decoration, is a colorless transparent liquid with three isomers: ortho, meta, and para. It has a flash point of 31.1°C and an explosion limit of 11%—64%. It is slightly soluble in water and belongs to the class of low-toxicity low-molecular-weight organic solvents. The main uses of xylene solvent include: as organic solvents for industrial use in paints, coatings, pigments, printing and dyeing; as softeners and solvents for plastics, rubber, etc.; as solvents for adhesives, cellulose, coatings, printing inks, etc.; for cleaning electronic equipment and components; and for analytical chemistry, environmental protection, and other purposes. In this article, you can learn more about the side effects, uses, differences from acetone, and how to use xylene solvent safely.
Xylene solvent is a colorless transparent liquid composed of xylene, a product of the substitution of two hydrogen atoms on toluene by methyl groups. It is an important organic chemical raw material and classified as a toxic substance according to the Major Hazard Identification (GB18218-2000).
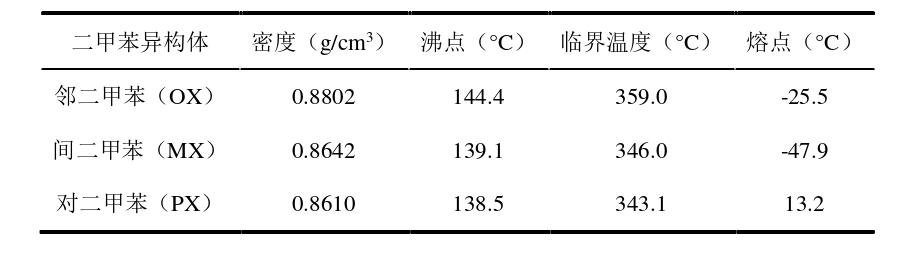
The molecular formula of xylene is C8H10. As a colorless transparent aromatic hydrocarbon compound, it is essentially formed by the substitution of two hydrogen atoms on the benzene ring in ortho, meta, and para positions with methyl groups. All three isomers of xylene are slightly irritating liquids with a characteristic odor at room temperature. Although insoluble in water, they can be mixed with various organic solvents such as anhydrous ethanol, ether, and chloroform. The main physical properties of the three isomers of xylene are shown in the table below. The main physical properties of the three isomers of xylene are very similar, especially the boiling point, with a maximum temperature difference of only 5.9°C.
The applications of xylene solvent are extensive, used as solvents in industries such as coatings, resins, dyes, and inks, as well as in the synthesis of monomers or solvents in pharmaceuticals, explosives, pesticides, etc. Furthermore, due to its solubility in organic solvents, xylene solvent is also widely used in the fields of PX and blending. In the chemical industry, rubber, and other fields, xylene solvent also plays a crucial role as an organic solvent, reaction intermediate, inhibitor, and additive.
Acetone is a colorless transparent liquid composed of two carbon atoms and one oxygen atom, with the chemical formula C3H6O. Acetone has a melting point of -94.9°C, a boiling point of 79.6°C, and a density of 0.806g/mL. It has good solubility and can react with most organic and inorganic substances, thus widely used in chemical, pharmaceutical, agricultural, and other fields. Its toxicity is relatively low.
Chemically, both acetone and xylene are commonly used organic solvents, but xylene is composed of a benzene ring and methyl groups, while acetone consists of two carbon atoms and one oxygen atom. This difference may affect their ability to dissolve certain substances in specific applications.
In terms of solubility, xylene has strong solubility and can dissolve various substances, while acetone also has strong solubility but is more suitable for dissolving some non-polar substances. Therefore, in practical applications, if various substances need to be dissolved, xylene may be preferred, while acetone may be preferred for dissolving non-polar substances.
In terms of odor, acetone has a strong pungent odor, which may be irritating to the human body, while xylene has a relatively milder odor, which is more acceptable.
In terms of safety, acetone has low toxicity, while xylene has higher toxicity. The use of xylene requires strict compliance with safety procedures, while acetone may relax safety measures to some extent. In some special applications, such as medical and laboratory fields, acetone may be preferred.
Therefore, in practical applications, the choice of which solvent to use should be based on specific application scenarios and requirements to achieve the best results and safety. At the same time, attention should be paid to safe operation and storage to avoid harm to the environment and human health.
Xylene solvent is a common organic solvent with numerous applications. Based on the physical properties of xylene, such as colorlessness, transparency, aromatic odor, and high density, it is widely used in industries such as coatings, printing inks, and food-grade plastics.
Xylene solvent is widely used in the coatings industry as an effective solvent for various types of coatings. In industrial production, xylene solvent is often used to disperse pigments, resins, and other materials to form stable coating layers. Additionally, xylene solvent has good volatility, making coatings easy to dry, facilitating production and use. In coating production, common solvents include esters, alcohols, benzenes, ketones, and water-based solvents. Among them, benzene solvents are inexpensive but highly toxic. Xylene, due to its good solubility, stability, and low toxicity, has become one of the most popular solvents.
Xylene solvent also has extensive applications in the printing ink industry. During the printing process, xylene solvent is used to dissolve ink and apply it to the printing substrate. The low volatility of xylene solvent gives ink good light and water resistance, maintaining long-lasting gloss and color on printed materials. Xylene solvent can also improve the flow of ink, making printed materials clearer and more aesthetically pleasing. In gravure printing ink solvents, esters and alcohols have fruity odors but are expensive, while xylene solvent is relatively inexpensive. Therefore, it is widely used in most printing ink production.
Xylene solvent also finds applications in the pharmaceutical industry. For example, it is used in pharmaceutical factory production and operations to dissolve and mix various chemical raw materials and preparations, such as medicinal starch, gelatin, polyethylene glycol, etc. In the pharmaceutical industry, the low toxicity and high purity of xylene solvent are essential because they directly relate to the quality and safety of drugs.
Xylene solvent is a colorless transparent liquid with a strong irritating odor, flammable, and harmful to both human health and the environment. Ingestion or contact with eyes and skin may cause reversible liver and kidney damage and neurological disorders. Therefore, strict safety measures and precautions must be taken when handling xylene solvent.
The safety and environmental impact of xylene solvent are important issues to consider. Taking appropriate preventive measures and operating procedures can minimize potential health hazards and protect the environment from pollution:
A. Proper masks and gloves must be worn to avoid inhalation or contact with xylene. Strictly follow operating procedures in well-ventilated areas, and ensure proper disposal of all waste and residues to prevent environmental contamination.
B. Proper storage and handling are also critical to preventing side effects of xylene solvent. Xylene solvent should be stored in a safe environment, away from direct sunlight and high temperatures, and kept separate from other substances. Regular inspections should be carried out during storage to ensure the safety of the solvent.
C. In case of xylene solvent leakage, necessary protective measures, such as wearing a gas mask, should be taken immediately, and the contaminated area should be cleaned up as soon as possible. Pay special attention to avoiding human injury or environmental pollution during storage and handling of the solvent.
Choosing the right xylene solvent is crucial as it directly affects the quality and performance of products. Factors to consider include:
Xylene solvents can come in various types and grades, including pure xylene, xylene with additives, and xylene blends. Therefore, choose the solvent type and grade that suits your application needs. For industries such as medicine, explosives, and pesticides, special purity and compatibility are required.
Purity is essential for the quality of xylene solvent. The higher the quality of xylene solvent, the stronger its chemical reactivity and stability. Therefore, choosing a high-purity solvent is crucial for product quality. If you have special purity requirements, you can consult reputable suppliers and brands recommended by us.
The compatibility of xylene solvent is another important consideration. Compatibility refers to whether the solvent can be compatible with other components of the product. If xylene solvent is incompatible with other components of the product, it may lead to decreased performance or deterioration. Therefore, when choosing xylene solvent, confirm its compatibility with other components of the product.
Choosing reputable suppliers and brands is crucial. Reputable suppliers and brands typically provide high-quality xylene solvent with guaranteed after-sales service and quality assurance.
This article has introduced the uses, side effects, and differences from acetone of xylene solvent. Xylene solvent is a colorless transparent low-toxic substance mainly used in manufacturing dyes, fragrances, synthetic fibers, widely used as solvents and diluents in paints, rubbers, as well as essential components in household and industrial cleaners, degreasers, and paint strippers, and as coupling agents and active solvents in water-based coatings. However, xylene has certain irritability; it may cause central nervous system anesthesia and chronic poisoning, and also cause environmental pollution. Therefore, we cannot ignore its potential threats to our health and the environment.
Let's pay attention to the use of xylene solvent, utilize and control its potential risks scientifically, and let technology benefit humanity!
[1] Liu Ying, Zheng Fang, Yang Qiwei, et al. Research progress on adsorption and separation of xylene isomers [J/OL]. Acta Chemical Engineering Sinica, 1-19[2024-04-02]. http://kns.cnki. net/kcms/detail/11.1946.TQ.20240226.1301.013.html.
[2] Xiong Xianjin. Prediction of solid-liquid phase equilibrium data of multi-component systems related to p-xylene crystallization [J]. Synthetic Fiber Industry, 2023, 46 (06): 49-54.
[3] Weekend, Gai Yueting, Yue Xin, et al. Performance study of MCM-22 molecular sieve for xylene isomerization [J]. Petroleum Refining and Chemical Engineering, 2023, 54 (11): 47-52.
[4] Tian Chong, Sang Shengli, Zhang Hao, et al. Research on application of top temperature control system in xylene distillation process [J]. Journal of Jilin Institute of Chemical Technology, 2023, 40 (09): 59-64. DOI:10.16039/ j.cnki.cn22-1249.2023.09.011.
[5] Wang Qi, Yan Huaiyu, Sun Zhongyan, et al. Determination of three xylene isomers in sanitary napkins by headspace gas chromatography-mass spectrometry [J]. Shandong Textile Technology, 2023, 64 (04): 25- 28.
 |
 |
 |