In daily life, have you ever heard these situations:
"What to do when you catch a cold and have a fever?" "Take cephalosporin!"
"What to do with cough and sore throat?" "Take cephalosporin!"
"What if you have a bit of diarrhea?" "Take cephalosporin!"...
It seems cephalosporin has become a miracle drug that cures all diseases in people's daily life. Could it really be so magical?
Let's take this opportunity to talk about this antibiotic that has been on the front line of clinical use for 60 years — cephalosporin.
In 1812, Napoleon assembled an army of 570,000 to invade Russia but ultimately retreated from Moscow due to an outbreak of typhus. What is typhus? It is an acute intestinal infectious disease caused by the typhus bacterium, mainly transmitted through contaminated water or food via the fecal-oral route. Typical symptoms include persistent high fever, abdominal pain, and in severe cases, intestinal perforation or septicemia.
At the end of World War II, many cities in Italy experienced typhus epidemics due to poor sanitation conditions. However, in Cagliari, southern Sardinia, a baffling scene appeared.
There was a sewage river leading to the Susigiu Sea area. Although residents along the river often swam, played, and even caught and ate raw fish from the river, very few people fell ill. It seemed as if a divine magic had been cast on this area, keeping typhus at bay.
This abnormal phenomenon immediately attracted the attention of Professor Giuseppe Brotzu, a medical professor at the University of Cagliari. Inspired by previous cases involving penicillin, he immediately realized that mysterious microbes capable of producing antibiotics might be hidden in this sewage ditch. Brotzu began culturing the river water on agar medium and eventually obtained a strain of Cephalosporium mold. Tests confirmed that this Cephalosporium could secrete a substance that fought the typhus bacterium, exciting Brotzu immensely. But his joy was soon dampened — the substance was extremely unstable.
For a long time afterward, Brotzu tried many methods to isolate this unstable antibacterial component but failed repeatedly. After three hard years, his funds ran out, forcing the suspension of the experiments.

Figure 1. Brotzu and his paper Ricerche su di un nuovo antibiotico
Unwilling to stop, Brotzu published his research results in a local journal in Cagliari to attract other researchers' attention. He also compiled relevant data and handed it to an Allied military doctor on Sardinia. After reviewing the information, the doctor consulted the British Medical Research Council and eventually connected with Howard Walter Florey, who had won the Nobel Prize in Physiology or Medicine for penicillin research. Florey showed great interest, contacted Brotzu, and requested some Cephalosporium strains.

Figure 2. Florey
With more professional personnel and funding support, Florey's team successfully purified three cephalosporin compounds in 1954: cephalosporin P, N, and C. Among them, cephalosporin C was effective against penicillin-resistant bacteria, which attracted great attention from the team.
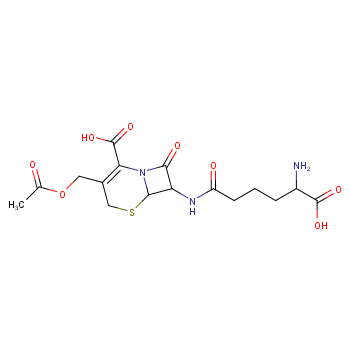
Figure 3. Structure of cephalosporin C
Later, researchers discovered that removing the side chain from cephalosporin C yielded a magical nucleus — 7-Aminocephalosporanic acid (7-ACA). Although this nucleus had weak antibacterial activity, it could produce a series of powerful cephalosporin antibiotics by adding different side chains. This opened the door to a new world, with various cephalosporin antibiotics emerging like mushrooms after rain.
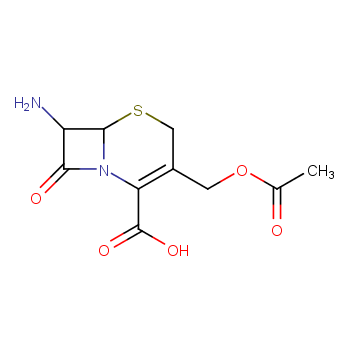
Figure 4. 7-ACA
Since the first cephalosporin drug, cephalosporin Thienamycin, was launched in 1964, the cephalosporin family has continued to grow and now spans five generations.

Figure 5. Classification of cephalosporin drugs
So how does cephalosporin work in the human body? We know that bacteria mainly consist of four basic structures: cell wall, cell membrane, cytoplasm, and nucleoid. Cephalosporins specifically bind to penicillin-binding proteins (PBPs) on bacterial cell membranes, inhibit transpeptidase activity, block peptidoglycan synthesis, interfere with and prevent bacterial cell wall formation, thereby destroying structural integrity and ultimately causing bacterial lysis and death.
Cephalosporins also have weaknesses. Their lifeline lies in the β-lactam ring in their structure (the red part in Figure 4). Once this ring is destroyed, cephalosporins lose their effectiveness. To resist cephalosporin attack, bacteria secrete various β-lactamases that destroy the β-lactam ring. As the saying goes, “The higher the magic, the higher the countermeasure.” Pharmacologists continuously introduce bulky groups into cephalosporin side chains to hinder β-lactamase from breaking the β-lactam ring. This explains why higher generation cephalosporins are more stable against enzymes than lower generation ones.
Does this mean the newer, higher generation, and more expensive cephalosporins always work better? Indeed, for Gram-negative bacilli, especially enzyme-producing resistant strains causing severe infections, third- and fourth-generation cephalosporins are clearly more effective than the first and second generations. However, for diseases caused by Gram-positive bacteria like Staphylococcus aureus, such as skin and soft tissue infections and upper respiratory infections, the efficacy of third-generation cephalosporins is inferior to the first and second generations. Moreover, in community-acquired infections, resistant bacteria are relatively rare. Even for Gram-negative infections, second-generation cephalosporins can cover the antibacterial spectrum well, making higher generation antibiotics unnecessary. This reminds us to emphasize targeted treatment and avoid blindly using higher generation cephalosporins regardless of disease severity, which not only wastes medicine but also induces bacterial resistance, causing more severe consequences.
Besides, we should pay attention to the following medication matters:
All medicines have side effects, so some patients stop taking cephalosporins once symptoms improve slightly. In fact, antibacterial drugs clear pathogens in a continuous and slow process. Stopping medication prematurely can cause relapse and promote bacterial resistance. Therefore, strict adherence to the prescribed course is essential.
As a member of the antibiotic family, cephalosporins have broken the limitations of penicillin, providing alternatives for penicillin-allergic patients, and expanding treatment options for children, the elderly, and immunocompromised individuals. From an unnoticed sewage river mold to a pillar of anti-infection treatment, cephalosporins have saved countless lives and reshaped the treatment landscape of infectious diseases. They have achieved generational upgrades in antibacterial therapy with lower toxicity, broader coverage, and stronger response to resistant bacteria. The strategy of developing new drugs through side chain modification has also provided a key technological paradigm for future antibiotic research.
 |