
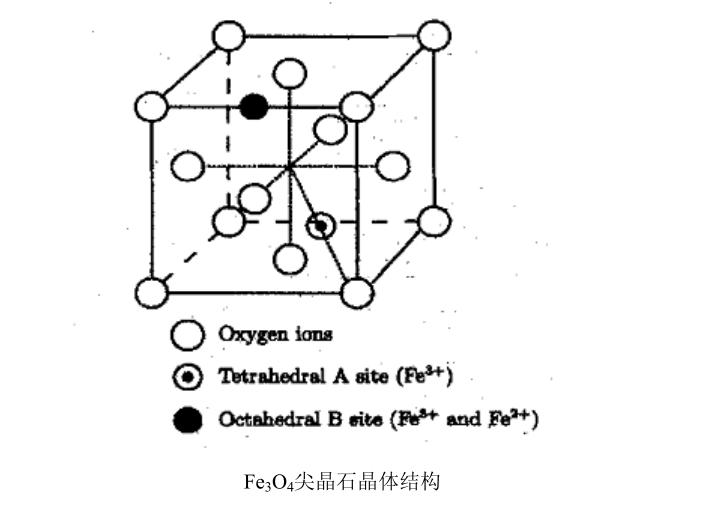
Black iron oxide, chemical formula Fe3O4, is an inorganic compound also known as iron(II,III) oxide. It exists with both iron in its stable oxidation states (+2 and +3), referred to as "magnetite" in mineralogy. Unlike hematite, it contains Fe2+ and Fe3+ ions simultaneously and appears as a black powder. Additionally, it exhibits ferromagnetism. The compound has a molar mass of 231.53 g/mol, melting point at 1,597°C, and boiling point at 2,623°C. Structurally, it adopts a cubic inverse spinel structure with cubic, close-packed oxide ions, where half of the Fe2+ ions occupy octahedral positions and Fe3+ ions are evenly distributed among the remaining octahedral and tetrahedral sites.
Red iron oxide, chemical formula Fe2O3, is iron oxide (III). It is the primary oxide of iron and known as "hematite" in mineralogy. It serves as a major source of iron in the steel industry and exhibits ferromagnetism. It has a molar mass of 159.69 g/mol and a melting point ranging approximately from 1,539 to 1,565°C, decomposing at higher temperatures. Therefore, it is insoluble in water. It exists in different structures termed as "polymorphs," such as α-phase and γ-phase, where each iron cation is coordinated by six oxygen ligands. Additionally, it has hydrated forms and appears as a reddish-brown solid, distinguishing it clearly from other iron oxides.
What is the difference between red iron oxide and black iron oxide? Red iron oxide, Fe2O3, and black iron oxide, Fe3O4, differ primarily in their chemical composition and appearance. Red iron oxide is a reddish-brown solid, whereas black iron oxide is a black solid powder. Red iron oxide is magnetically ferromagnetic, whereas black iron oxide exhibits ferrimagnetism.
Synthesized red iron oxide pigments are the most common and highest in iron content among iron oxides. Red iron oxide is widely favored for its pure color, stability, and strong coloring properties. Characteristics include:
Fine particle size, strong coloring power
Non-toxic
Non-combustible
Alkali-resistant
Produces attractive shades
Additionally, they possess pure hues and consistent performance. Red iron oxide finds extensive use in paints, coatings, rubber, paper, cement pigments, and construction materials. The construction materials industry consumes the most iron oxide pigments due to their excellent dispersibility and coloring intensity. They are used for coloring concrete, mortar, paving stones, and bricks.
Synthesized black iron oxide serves as a source of iron in ceramic applications, crucially in achieving original black colors. The main difference between red and black iron oxide pigments lies in black iron oxide being ferrimagnetic. Characteristics include:
Inherently ferrimagnetic
Melting point at 1,597°C (2,907°F)
Boiling point at 2,623°C (4,753°F)
Conductivity, significantly higher than red iron oxide
Black iron oxide pigments also serve as colorants in various non-ceramic products. Additionally, these black oxides are used for corrosion protection in various appliances and are components in anti-corrosion coatings (used in many bridges).
Red iron oxide is produced by calcining black iron oxide particles in an oxidizing atmosphere. Subsequently, it undergoes jet milling to produce "micronized" red iron oxide particles with a particle size of approximately 325 mesh. This red iron oxide is highly heat-resistant, up to 1832°F (1000℃). In contrast, black iron oxide oxidizes at 365°F (180℃), changing from black to brown and eventually to red as particle size increases, transitioning from light pinkish-red to red and then deep purplish-red.
How to convert red iron oxide to black iron oxide? WO1999037719 discloses a method to produce black iron oxide pigment from Fe2O3 hematite iron oxide, which can be impure oxides produced via the "Ruthner" process or other methods, or mineral hematite. This method upgrades inferior synthetic or natural hematite by converting it into black pigment, subsequently oxidizing the resulting magnetite back into hematite to enhance its performance as a red pigment. The process involves a low-temperature aqueous slurry reaction where hematite contacts soluble Fe(II) ion sources in the presence of soluble Fe(III) ion sources in an alkaline aqueous medium to convert it into Fe3O4. This specific process occurs during boiling water reaction with hematite, where boiling water contacts a soluble Fe(II) ion source in the presence of a Fe(III) ion source, transforming it into Fe3O4.
What is a substitute for black iron oxide? Depending on the application, suitable substitutes for black iron oxide may include:
Black pigments such as carbon black or manganese dioxide can substitute black iron oxide in applications like paints, plastics, and coatings.
For specific magnetic needs, materials like magnetite nanoparticles or synthetic spinels can substitute black iron oxide.
Depending on the specific catalytic reaction, other metal oxides or even non-metallic catalysts may be viable alternatives.
Red and black iron oxides exhibit significant differences in color, chemical properties, and application areas. Understanding their characteristics helps in making the right choices for practical applications to meet various needs and requirements. Comparative studies enable better utilization of these two pigments, providing more effective solutions across industries.
[1] https://www.differencebetween.com/difference-between-red-and-black-iron-oxide/
[2] https://bansaltrading.com/usage-of-synthetic-iron-oxide
[3] https://patentscope.wipo.int/search/en/detail.jsf?docId=WO1999037719
[4] https://reade.com/product/magnetite-black-iron-oxide/
[5] https://concreteproducts.com/index.php/2022/02/07/manganese-ferrite-pigment-carbon-black-iron-oxide-alternative/
 |
 |
 |