Cis-trans isomerism is a type of stereoisomerism caused by the inability of the two carbon atoms connected by a double bond to rotate freely around the sigma bond. This typically refers to alkenes, but can also apply to C=N double bonds, N=N double bonds, and cyclic compounds.
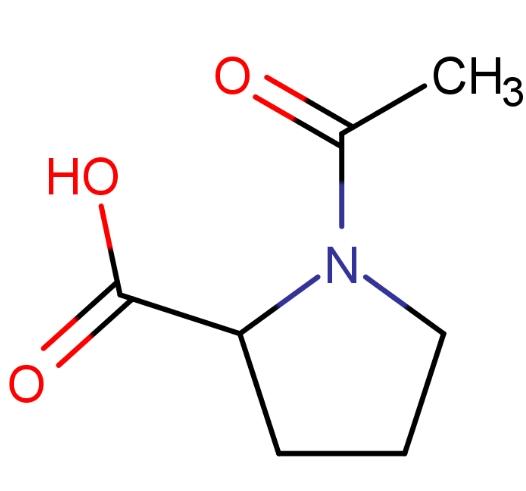
Below are the cis and trans isomers of N-acetyl-L-proline:
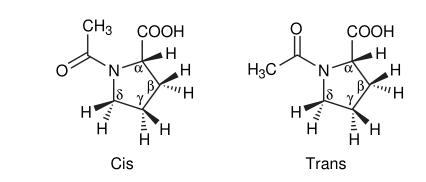
In proteins, the delocalization of electrons in the amide bond between adjacent amino acid residues restricts the rotation of the bond between nitrogen and carbonyl carbon. Therefore, the four atoms in the peptide group (O–C–N–X, with X being H for proline) and the adjacent alpha carbon are coplanar or nearly coplanar. Over 99% of peptide bonds adopt the trans configuration, keeping the alpha carbon and its large side chain groups on the opposite side of the C–N bond. However, in proline amide bonds, X is the δ-carbon of the pyrrolidine ring, introducing additional spatial constraints. This results in the cis and trans isomers having nearly equivalent energy. Thus, both isomers are observed in natural systems, k trans of n-acetyl-l-proline:
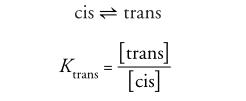
The cis-trans isomeric equilibrium of N-acetyl-L-proline is a simple yet significant system that clearly demonstrates the effects of solvent polarity and solution pH on its equilibrium. This research is valuable not only for biophysical experiments but also for instrumental analysis and organic spectroscopy labs.
Kathryn R. Williams and colleagues studied the impact of solvent pH and polarity on the Ktrans equilibrium constant of the cis and trans isomers of N-acetyl-L-proline using hydrogen nuclear magnetic resonance (H-NMR). ktrans values for n-acetyl-l-proline, an indicator of the balance between cis and trans isomers, is determined by integrating the areas under peaks in the H-NMR spectra. This value helps determine the relative amount of cis isomer in the sample.
N-acetyl-L-proline (N-AcPro) serves as a simple model for proline peptide bonds. Extensive NMR studies on this equilibrium have been reported in various solvent systems.
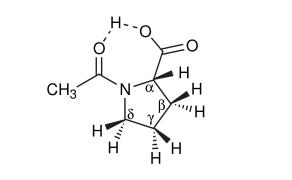
Studies observed polarity effects by determining Ktrans in d6-acetone (relatively polar) and a 45:55 d6-benzene/CDCl3 mixture (non-polar). Four solvent systems (45:55 d6-benzene/CDCl3, d6-acetone, pH 7 D2O) demonstrated the effects of solvent polarity and water phase pH. Given that peptide bonds of interest primarily occur in aqueous environments, Ktrans of N-AcPro was also measured in D2O at two pH levels (below 2 and above 7). Previous research indicated that the cis isomer is more acidic (with a pKa approximately 0.6 units lower). Additionally, when N-AcPro is fully deprotonated, the cis-trans equilibrium shifts slightly towards the cis isomer (Ktrans approximately 0.8). These observations initially seemed consistent with protonated trans isomers having intramolecular hydrogen bonding. However, Lauterwein et al. measured the 17O chemical shifts of N-AcPro across pH transitions and observed parallel behavior between cis and trans forms. The pKa, chemical shifts, and Ktrans effects were attributed to the orientation of the amide dipole relative to the carboxyl group. The chosen pH values were meant to clearly display the impact of pH on Ktrans. The following figure shows the Hα and Hδ regions of N-acetyl-L-proline in a benzene-chloroform mixture. Similar resolution was observed in other solvent systems. Students assessed Ktrans values by integrating four groups (α-trans vs. α-cis, δ-trans vs. δ-cis, α-trans vs. δ-cis, and δ-trans vs. α-cis) and calculated 95% confidence intervals.

As shown in the table of typical data, the effects of polarity and pH are clearly demonstrated:
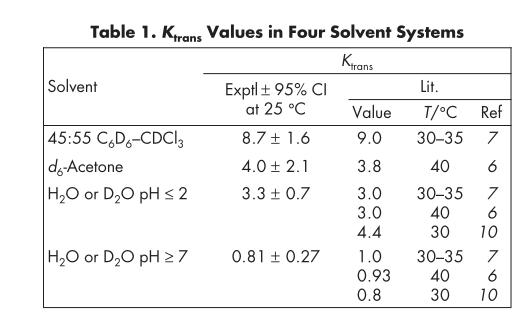
Aside from high pH aqueous solutions, the trans isomer predominates. In non-aqueous systems, the equilibrium constant increases with decreasing solvent polarity. It is generally believed that in non-polar solvents, the trans isomer benefits from forming intramolecular hydrogen bonds, which "protect" the carboxyl hydrogen from the non-polar environment.
The preference for the trans isomer of N-acetyl-L-proline under low pH conditions relates to the impact of pH on intramolecular hydrogen bonds and electrostatic interactions. At low pH, a high concentration of hydrogen ions (H+) binds to the carboxyl hydrogen atoms of the proline molecule, reducing the negative charge on the carboxyl group. This change eliminates interactions caused by electrostatic repulsion between carboxyl groups. Specifically, the trans isomer is more stable due to stronger intramolecular hydrogen bonds between the carboxyl and amide oxygen atoms, reducing internal steric hindrance. This hydrogen bonding stabilizes the trans isomer in non-polar solvents, as the hydrogen bonds lower the polarity of the molecule and decrease electrostatic interactions. In low pH conditions, the hydrogen atoms on the carboxyl group are not removed, avoiding steric hindrance from electrostatic repulsion, thus enhancing the stability of the trans isomer. Conversely, in high pH environments, basic conditions remove hydrogen atoms from the carboxyl group, causing electrostatic repulsion within the molecule and leading to instability of the trans isomer and a shift towards the cis isomer. Therefore, N-acetyl-L-proline is more likely to form the trans isomer in low pH environments.
 |
 |
 |