
In agricultural production, deltamethrin vs flumethrin are both common insecticides used to control pests and improve crop yield and quality. Despite both belonging to the pyrethroid class of insecticides, they differ in chemical structure, insecticidal properties, and their impact on the environment and human health. This article will compare deltamethrin and flumethrin to help readers better understand the differences between these two insecticides, aiding in making more scientifically informed choices in practical applications.
Flumethrin, also known by its English name "flumethrin", CAS number: 69770-45-2, molecular formula: C28H22Cl2FNO3, appearance and properties: a transparent brown liquid, density: 1.342 g/cm3, refractive index: 1.629. Flumethrin is a type of pyrethroid insecticide. It is used in veterinary medicine to combat parasitic insects and ticks on cattle, sheep, goats, horses, and dogs, as well as to treat parasitic mites in bee colonies.
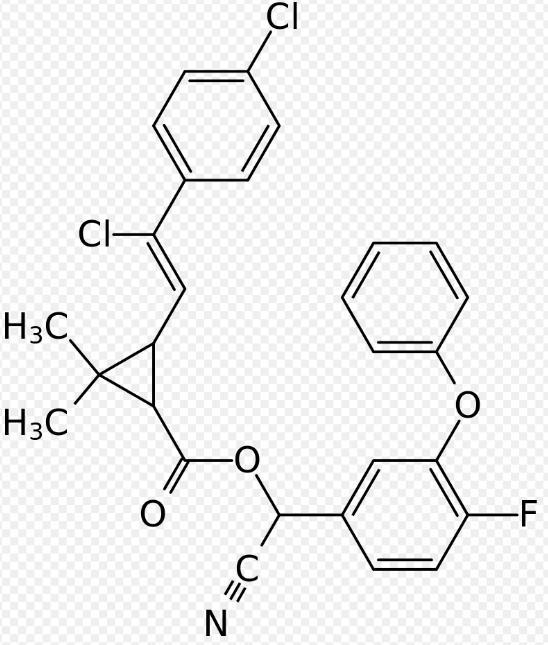
Deltamethrin, also known by its English name "deltamethrin", CAS number: 52918-63-5, molecular formula: C22H19Br2NO3, appearance and properties: white to grayish-white crystals or powder, density: 1.595g/cm3, refractive index: 1.653, boiling point: 300°C, melting point: 98°C. Deltamethrin is a type of pyrethroid ester insecticide. It plays a crucial role in controlling malaria vectors and is used in the manufacture of long-lasting insecticidal mosquito nets; however, mosquitoes and bedbugs have developed widespread resistance to deltamethrin.
Deltamethrin is toxic to aquatic organisms, particularly fish. While generally considered safe for use around humans, it still possesses neurotoxic properties. It is an allergen that can trigger asthma in certain individuals.
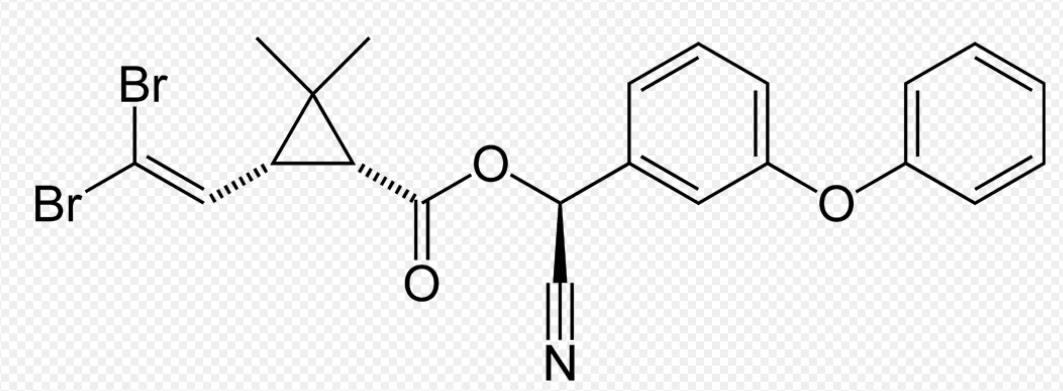
Deltamethrin and flumethrin both belong to the pyrethroid class of insecticides. They disrupt the nervous systems of insects, leading to paralysis and death. Both insecticides act on the voltage-gated sodium channels in insect nerve cells. Their specific actions include:
Deltamethrin vs flumethrin bind to sodium channels, preventing them from closing normally after opening. This disrupts the normal flow of sodium ions across nerve cell membranes, leading to uncontrolled nerve impulses.
The sustained firing of nerve impulses overwhelms the nervous system, causing hyperexcitation, tremors, paralysis, and eventual insect death.
Second-generation photostable pyrethroids emerged in the 1970s. They exhibit residual activity (several weeks) on skin and in the environment. The major second-generation pyrethroids are cypermethrin, cyhalothrin, permethrin, deltamethrin, and flumethrin.
They act upon contact with insects and mites, and their mode of action is very similar to that of organochlorines: they open Na+ channels, inducing nerve cell depolarization. The rapid action on insect brain ganglia leads to sudden insect shock, termed the "knockdown" effect. Insects cease all movement, behaving as if dead. This knockdown is reversible, and within seconds, insects may wake up and enter a second phase, which involves rapid, brief, and inconsistent movements due to hyperexcitation of peripheral nerves, which may lead to death. The spectrum of activity varies with the molecule. Cypermethrin and deltamethrin are both insecticides and acaricides. These molecules are volatile, and their presence around treated animals explains their repellent action on flying insects (mosquitoes, sandflies) and even ticks. This repellency is a major advantage of the pyrethroids recently developed for dogs.
In flea collars, deltamethrin is one of the safest insecticides. Deltamethrin is classified as a synthetic pyrethroid, meaning it is a synthetic derivative of natural insecticide pyrethrins, which are extracts found in chrysanthemum flowers; flumethrin is a popular insecticide mainly acting by disrupting the nervous systems of ticks and biting flies. Flumethrin works best when used in combination with other ingredients.
Deltamethrin and flumethrin are both second-generation synthetic pyrethroids. Type II synthetic pyrethroids contain an α-cyano group, which induces a syndrome in rodents characterized by pawing, burrowing, salivation, hypothermia, and coarse tremors leading to choreoathetosis (CS syndrome). The common adverse outcome pathway (AOP) for synthetic pyrethroids involves their ability to interact with voltage-gated sodium channels (VGSC) in the central and peripheral nervous systems, leading to altered neuronal firing and eventual neurotoxicity. Clinical symptoms characteristic of type II synthetic pyrethroids, such as increased salivation, activity/gait changes, and tremors, are observed in experimental toxicology studies, including acute and subchronic neurotoxicity studies in rats, subchronic and chronic studies in dogs and rats, and developmental and reproductive studies in rats. Besides these clinical symptoms, increased sensitivity to external stimuli, altered vocalization, and decreased grip strength in forelimbs and hindlimbs are commonly observed in databases.
In pre-natal developmental studies in rats and rabbits, deltamethrin did not exhibit any adverse effects on fetuses or offspring; thus, there was no quantitative or qualitative evidence of susceptibility in these studies. However, qualitative susceptibility to high doses was observed in developmental neurotoxicity (DNT) and second-generation reproductive studies, as effects on offspring were more severe than those on dams. In DNT studies, an increase in the incidence of vocalization during the functional observational battery (FOB) was observed in male pups on postnatal day (PND) 4 of treatment, and reduced body weight was observed in both sexes pre- and post-weaning. In maternal animals, only body weight reduction was observed, although neurological measurements similar to those in pups, including FOB analysis, were conducted; no adverse FOB effects were observed. In second-generation reproduction studies, treatment-related effects in high-dose parental animals were limited to lesions in the head, neck, or forelimbs, alopecia in males, and ataxia and hyper-reactivity during pregnancy in females. Increased mortality in offspring (PND 8-14) and early clinical signs post-weaning (i.e., impaired righting reflex, hyperactivity, limb splaying, vocalization, and excessive salivation) were observed at high doses in F1 offspring, with no increase in mortality or clinical signs in F2 offspring. Reduced body weights were observed in adults of the P and F1 generations, with reduced body weights in F1 and F2 pups.
Flumethrin blocks the closure of sodium channel ion gates during repolarization of nerve cell membranes. This severely disrupts the transmission of nerve impulses, leading to spontaneous depolarization or repetitive firing of membranes. At low concentrations, insects and other arthropods exhibit hyperactivity. At high concentrations, they become paralyzed and die. Sensory and nerve cells are particularly sensitive.
Flumethrin and pyriproxyfen are ingredients commonly found in flea and tick collars. While generally safe, minor side effects such as skin irritation, redness, and hair loss around the collar area may occur. These issues usually resolve within a few weeks. If your pet experiences severe irritation or persistent symptoms, remove the collar and consult your veterinarian.
Is flumethrin and permethrin the same? Both are insecticides used for flea and tick prevention, but they are not the same substance. Permethrin may be safer for cats than flumethrin in general.
Deltamethrin and flumethrin, as two common insecticides, play important roles in agricultural production. While both possess insecticidal properties, they differ in chemical structure, insecticidal characteristics, and safety profiles. Agricultural producers should consider factors such as specific crops, pest situations, and environmental protection when choosing to use them to avoid potential risks associated with improper use.
[1]https://www.federalregister.gov/documents/2023/04/04/2023-06939/deltamethrin-pesticide-tolerances
[2]https://en.wikipedia.org/wiki/
[3]https://drugs.ncats.io/drug/2O051W13LH
[4]https://www.sciencedirect.com/topics/medicine-and-dentistry/flumethrin
[5]https://tevrapet.com/are-flea-collars-right-for-your-dog/
[6]https://parasitesandvectors.biomedcentral.com/articles/10.1186/1756-3305-5-79
 |
 |
 |