High-performance liquid chromatography (HPLC) is a commonly used analytical technique in the pharmaceutical field, known for its high sensitivity, resolution, and efficiency. It is widely applied in drug content determination and quality control. Dextromethorphan hydrobromide is a commonly used antitussive medication, and accurate determination of its content is crucial for ensuring drug quality and safety.
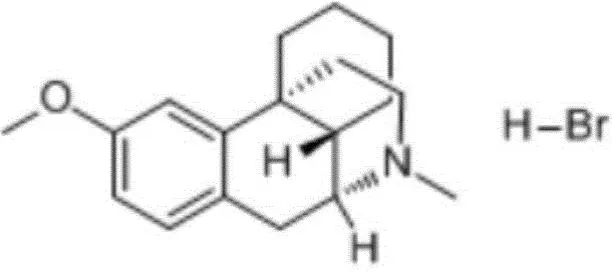
Overview: Dextromethorphan hydrobromide, chemically known as 3-methoxy-17-methyl-(9α,13α,14α)-morphinan hydrobromide, with CAS number 125-69-9, has the following structure:
Dextromethorphan hydrobromide is commonly used as a central nervous system cough suppressant, mainly for dry coughs. It is suitable for coughs associated with colds, acute or chronic bronchitis, bronchial asthma, pharyngitis, pulmonary tuberculosis, and other upper respiratory tract infections. Existing formulations of dextromethorphan hydrobromide include tablets, capsules, injections, and oral solutions.
Determination of dextromethorphan hydrobromide content in medications:
(1) Report One
Lixing et al. established a high-performance liquid chromatography method for simultaneous determination of norepinephrine hydrochloride, chlorpheniramine maleate, and dextromethorphan hydrobromide in cough oral solutions. An ACE Excel super C18 column (250 mm × 4.6 mm, 5μm) was used for separation, with mobile phase A as potassium dihydrogen phosphate and sodium octanesulfonate buffer-methanol solution (volume ratio 92:8), and mobile phase B as potassium dihydrogen phosphate and sodium octanesulfonate buffer-methanol solution (volume ratio 12:88), gradient elution, flow rate of 1.0 mL/min, detection wavelength of 220 nm, column temperature of 35°C, and injection volume of 10μL.
The mass concentrations of norepinephrine hydrochloride, chlorpheniramine maleate, and dextromethorphan hydrobromide were linearly correlated with the corresponding chromatographic peak areas in the ranges of 38.876-64.793, 16.256-24.384, and 76.723-127.872μg/mL, respectively. The relative standard deviations of the three components' content determination results were 0.28%-0.42% (n=6), and the spiked recovery rates were 98.65%-99.65%. This method is simple, stable, reproducible, and suitable for simultaneous determination of norepinephrine hydrochloride, chlorpheniramine maleate, and dextromethorphan hydrobromide content in cough oral solutions.
(2) Report Two
Qiukexian et al. established a method for simultaneous determination of acetaminophen, dextromethorphan hydrobromide, and dextromethorphan hydrobromide-related substances in soft capsules using high-performance liquid chromatography (HPLC). The chromatographic column used was Phenomenex Luna C18 (250×4.6 mm, 5μm), with the mobile phase A as an aqueous solution of sodium dodecyl sulfate and ammonium acetate, and the mobile phase B as acetonitrile-acetic acid (2:1), gradient elution; detection wavelength: 280 nm; column temperature: 30°C.
The main components and known impurities showed good linear relationships with peak areas within the corresponding concentration ranges, with r=1.00; the average recovery rates were in the range of 91%-114%, and the relative standard deviations (RSDs) were all less than 10%. The quantitation limits (relative to the concentration levels of the test samples) were all below 0.05%. This method is simple, efficient, highly sensitive, and has good accuracy and precision, suitable for quality control of acetaminophen, dextromethorphan hydrobromide, and dextromethorphan hydrobromide-related substances in soft capsules.
(3) Report Three
Luyufeng et al. established an HPLC method for simultaneous determination of acetaminophen, pseudoephedrine hydrochloride, and dextromethorphan hydrobromide contents and uniformity in acetaminophen-pseudoephedrine hydrochloride-dextromethorphan hydrobromide dry mix suspensions. The chromatographic column used was Kromasil 100-5-C18 (4.6 mm×250 mm, 5 μm), with the mobile phase composed of a 0.05 mol·L-1 potassium dihydrogen phosphate solution (pH adjusted to 3.5 with phosphoric acid) and methanol, gradient elution; flow rate: 1.0 mL·min-1; detection wavelength: 215 nm; injection volume: 10 μL; column temperature: 30°C.
The linear ranges of acetaminophen, pseudoephedrine hydrochloride, and dextromethorphan hydrob
romide were 0.162-1.136 mg·mL-1 (r=0.9999), 15.05-105.35 μg·mL-1 (r=1.000), and 5.01-35.07 μg·mL-1 (r=0.9996), respectively. The average recovery rates at high, medium, and low concentrations were all >98% (n=3), with RSDs all <1.0% (n=3). This method is accurate, simple, sensitive, and suitable for simultaneous determination of the three components' content and uniformity in acetaminophen-pseudoephedrine hydrochloride-dextromethorphan hydrobromide dry mix suspensions, providing a reference for quality control of this product.
(4) Report Four
Wu Jin et al. established an HPLC method for determining the contents of acetaminophen, doxylamine succinate, and dextromethorphan hydrobromide in cold capsules. The method involved using an Agilent Zorbax C18 column (4.6 mm×150 mm, 5μm), with acetonitrile as mobile phase A and buffer solution (pH=3.0) as mobile phase B, gradient elution, flow rate of 1.2 ml/min, detection wavelength of 275 nm, column temperature of 35°C, and injection volume of 20μl.
The linear ranges of acetaminophen, doxylamine succinate, and dextromethorphan hydrobromide were 2.62-26.23 μg/ml (r=0.9999), 2.51-25.14 μg/ml (r=0.9998), and 5.68-56.78 μg/ml (r=0.9999), respectively. The recovery rates were 100.6%, 101.1%, and 100.2%, respectively, with RSDs of 0.4%, 1.2%, and 0.4% (n=9). This method is simple, rapid, accurate, and reliable for determining the contents of acetaminophen, doxylamine succinate, and dextromethorphan hydrobromide in cold capsules.
(5) Report Five
Jie Chunwen et al. developed an HPLC method for simultaneous determination of the main components (pseudoephedrine hydrochloride, chlorpheniramine maleate, and dextromethorphan hydrobromide) in a solution of antitussive, decongestant, and expectorant medications. The study used a Thermo C18 column (4.6mm×250mm, 10μm); the mobile phase consisted of a 0.02%mol.L-1 potassium dihydrogen phosphate solution (pH adjusted to 4.0±0.1 with phosphoric acid) - methanol (20:80) as mobile phase A and a 0.02%mol.L-1 potassium dihydrogen phosphate solution (pH adjusted to 4.0±0.1 with phosphoric acid) - methanol (55:45) as mobile phase B for gradient elution; flow rate: 1mL/min; detection wavelengths: 257nm (pseudoephedrine hydrochloride) and 264nm (chlorpheniramine maleate and dextromethorphan hydrobromide).
The linear range of pseudoephedrine hydrochloride was 0.9624-81.92496 mg.mL-1, with an average recovery rate of 99.67% and RSD of 0.83% (n=9); the linear range of chlorpheniramine maleate was 0.0645-60.12912 mg.mL-1, with an average recovery rate of 100.23% and RSD of 0.87% (n=9); the linear range of dextromethorphan hydrobromide was 0.3200-880.64176 mg.mL-1, with an average recovery rate of 99.70% and RSD of 0.93% (n=9). This method is accurate, simple, fast, and suitable for simultaneous determination of the three components in the solution of antitussive, decongestant, and expectorant medications.
References:
[1] Li Xin, Dai Minyan, Ning Xing, et al. Simultaneous determination of phenylephrine hydrochloride, chlorpheniramine maleate, and dextromethorphan hydrobromide in antitussive oral liquid by high performance liquid chromatography [J] . Chemical Analysis and Metrology, 2023, 32 (07): 70-73.
[2] Qiu Kexian, Liu Xiaofen. Determination of related substances in soft capsules containing acetaminophen, dextromethorphan hydrobromide and doxylamine succinate by high performance liquid chromatography [J]. Chinese Pharmacoeconomics, 2022, 17 (03): 121-124+128.
[3] Lu Yufeng, Li Jie. Simultaneous determination of content and content uniformity of three ingredients in acetaminophen Asami dry suspension by high performance liquid chromatography [J]. Central and Southern Pharmacy, 2020, 18 (11): 1906-1909.
[4] Wu Jin, Zhu Lisheng, Yu Li, et al. Determination of acetaminophen, doxylamine succinate, and dextromethorphan hydrobromide in cold soft capsules by high performance liquid chromatography [J]. Chinese Hospital Drugs Evaluation and Analysis, 2016, 16 (07): 956-958. DOI:10.14009/j.issn.1672-2124.2016.07.035.
[5] Jiangsu Hanchen Pharmaceutical Co., Ltd. A dextromethorphan hydrobromide oral solution. 2022-03-11.
[6] Xie Chunwen, Yang Shaohui, Shi Dayong. Determination of the contents of three components in Memin pseudoanesthetic solution by high performance liquid chromatography [J]. Strait Pharmacy, 2008, (07): 59-61.