Ethyl cinnamate is a common organic compound found widely in natural fruits, spices, and essential oils. It is also frequently used in food additives, flavorings, and cosmetics. However, in recent years, the potential ethyl cinnamate hazards have also raised concerns. This article will outline the ethyl cinnamate hazards, including its potential effects on the skin, respiratory system, and reproductive system. Additionally, it will discuss precautions when handling ethyl cinnamate and how to mitigate related risks.
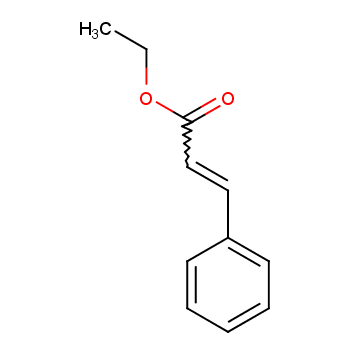
Ethyl cinnamate, also known as β-phenylacrylic acid ethyl ester (molecular formula: Ph CH=CHCOOEt), is present in natural tonka beans and has a fruity floral aroma. It is suitable for formulating daily fragrances such as rose, jasmine, narcissus, gardenia, cologne, jonquil, and citrus. Additionally, it is used in preparing perfume essences, soap essences, and as a fixative in perfumery.
According to the 2012 OSHA Hazard Communication Standard (29 CFR 1910.1200), this chemical is not considered hazardous.
It causes slight dangers in acute inhalation studies in mice, leading to drowsiness and changes in motor activity; in rats, it causes blood changes and enzyme effects in 17-week intermittent oral studies [RTECS]; its use as a flavoring agent in food is safe [JECFA]; it may cause irritation [Alfa Aesar MSDS].
Skin contact (irritation, permeator), eye contact (irritation), ingestion, and inhalation pose slight risks.
Carcinogenicity: Unknown.
Mutagenicity: Not available.
Teratogenicity: None.
Developmental toxicity: None.
The aggravation of symptoms with repeated or prolonged exposure is currently unclear.
Immediately flush with plenty of water, also under eyelids, for at least 15 minutes.
Immediately wash with soap and plenty of water while removing all contaminated clothing and shoes. Consult a doctor.
Remove from exposure, lie down. Move to fresh air. If breathing is difficult, give artificial respiration. Consult a doctor.
Rinse mouth with water. Consult a doctor.
Avoid skin and eye contact. Avoid contact with skin and clothing. Remove contaminated clothing and gloves, including the inside, and wash before reuse. Avoid inhalation of vapors or mists. Do not ingest. If ingested, seek medical assistance immediately. Thoroughly clean after handling.
Keep container tightly closed in a dry, cool, well-ventilated place. Incompatible materials. Acid. Reagents. Oxidants.
Data on the impact of ethyl cinnamate on terrestrial ecosystems are limited. In aquatic organisms, studies have shown that ethyl cinnamate may be toxic to aquatic organisms, especially algae. At relatively low concentrations, it disrupts the photosynthesis of algae, which may affect the base of the aquatic food chain.
Based on chlorophyll fluorescence and flow cytometry analysis, Yang Jiao et al. studied the toxic effects of ethyl cinnamate on the photosynthesis and physiological characteristics of Chlorella pyrenoidosa. Measurement indicators include biomass, maximum photochemical efficiency (F v/F m), actual photochemical efficiency (ФPSII), FDA, and PI staining fluorescence.
Yang Jiao's research results indicate: (1) With increasing exposure concentration, the inhibitory effect on biomass increases. 1 mg/L ethyl cinnamate is sufficient to reduce the total biomass of Chlorella pyrenoidosa. The EC50 values at 48 h and 72 h were 2.07 mg/L (1.94 ~ 2.20) and 1.89 mg/L (1.82 ~ 1.97), respectively. The effect of ethyl cinnamate on the biomass of Chlorella pyrenoidosa is as follows:

(2) After treatment with 2 ~ 4 mg/L ethyl cinnamate for 24 h, the photosynthesis of Chlorella pyrenoidosa almost ceases, manifested as ФPSII approaching zero. After 72 h of treatment with 4 mg/L ethyl cinnamate, F v/F m decreased to 0. (3) Ethyl cinnamate also affected the cellular physiology of Chlorella pyrenoidosa, but these effects resulted in the inhibition of cell yield rather than cell death. Ethyl cinnamate exposure reduced esterase activity, increased average cell size, and altered chlorophyll a fluorescence intensity. Overall, esterase activity was the most sensitive variable.
Although specific details of the degradation pathways are lacking, safety data indicate that ethyl cinnamate is not classified as environmentally harmful. This suggests that it may decompose at a reasonable rate in the environment.
Ethyl cinnamate is currently not classified as a hazardous substance by major regulatory agencies such as the International Agency for Research on Cancer (IARC), ACGIH, NTP, or OSHA. However, regulations regarding its use may vary depending on the application. Here are some relevant international resources: EU (Europe): The European Chemicals Agency (ECHA) retains registration of ethyl cinnamate. While it is not listed as a Substance of Very High Concern (SVHC), you can find regulatory information related to Biocidal Products Regulation (BPR).
Although ethyl cinnamate has extensive applications, its potential hazards should not be overlooked. When using ethyl cinnamate, strict adherence to safety protocols, personal protective measures, and close monitoring of one's health status are essential. If any adverse symptoms occur after using ethyl cinnamate, discontinue use immediately and consult a doctor. For a more comprehensive understanding of the ethyl cinnamate hazards, it is recommended to refer to its SDS sheet for specific physicochemical properties, health hazards, storage, and disposal methods. You can also consult with professional chemical safety personnel or medical professionals for more specialized advice and recommendations.
[1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4458563/
[2] https://www.spectrumchemical.com/media/sds/E0090.PDF
[3] https://www.fishersci.com/store/msds?partNumber=AC150531000&countryCode=US&language=en
[4] https://pubchem.ncbi.nlm.nih.gov/compound/Ethyl-cinnamate
 |
 |
 |