FMOC-Ala-OH is a derivative of amino acid used in peptide synthesis, with its full name being 9-fluorenylmethoxycarbonyl-L-alanine. It serves to protect amino groups during solid-phase peptide synthesis, ensuring the assembly of peptide chains in the desired sequence.
Fmoc-L-alanine, also known as FMOC-Ala-OH, CAS: 35661-39-3, molecular formula: C18H17NO4, appearance: white to gray-white crystalline powder, boiling point: 544.1℃ (760 mmHg), melting point: 150-154℃. The fmoc-ala-oh molecular weight is 311.33.
Fmoc-Ala-OH H2O is the Fmoc-L-alanine monohydrate, a type of Fmoc-L-amino acid used in peptide synthesis. Fmoc-L-alanine is an L-alanine protected by Fmoc. L-alanine is used for the determination of glucose and alanine metabolism in diabetic patients. N-Fmoc-L-alanine may be used in proteomics research and solid-phase peptide synthesis techniques. Alanine is one of the simplest amino acids with a methyl side chain. This small side chain offers high flexibility when incorporated into a peptide chain. The Fmoc group is typically removed with a base, such as pyridine, which is an orthogonally deprotecting strategy to the acid-labile Boc group. FMOC-Ala-OH?H2O is a type of Fmoc-L-amino acid used in peptide synthesis.
Na2CO3 (29.8 g, 280.9 mmol) and 9-fluorenylmethyl-N-succinimidyl carbonate (28.4 g, 84.27 mmol) were added to a solution of L-alanine (5 g, 56.18 mmol) in water/dioxane (volume ratio 1:1, total 400 mL) and stirred at room temperature for 18 hours. After the reaction, the reaction mixture was filtered, and the organic layer was extracted with 100 mL × 3 of ether. The aqueous phase was acidified with concentrated hydrochloric acid and extracted with 150 mL × 3 of ethyl acetate. After combining the organic phases, the mixture was dried over Na2SO4 and concentrated to yield 17.3 g of a white solid, with a yield of 99%.
FMOC-Ala-OH is a versatile reagent used in Fmoc solid-phase peptide synthesis. Common applications of FMOC-Ala-OH include:
FMOC-Ala-OH is a key reagent for Fmoc solid-phase peptide synthesis (SPPS). SPPS is a powerful technique for manufacturing peptides, amino acid chains that play crucial roles in biological processes. By serving as a building block, FMOC-Ala-OH enables researchers to incorporate the amino acid L-alanine into the peptide sequences they desire.
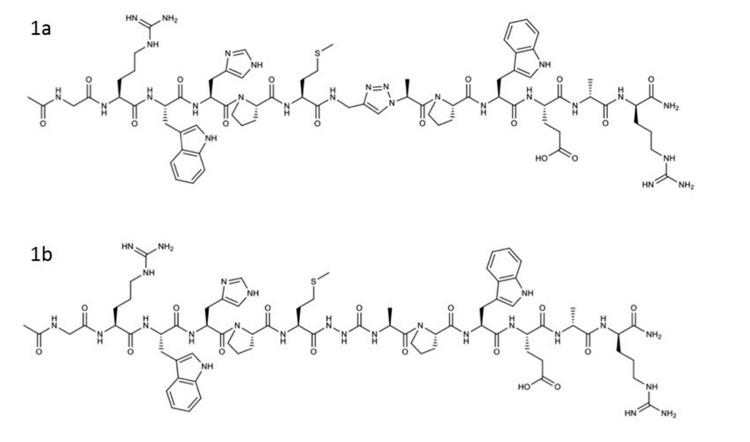
In the report by Matthieu Galibert et al., triazole peptides 1a and 2a and nitrogen heterocycles 1b and 2b (as shown in the figure below) were synthesized via Fmoc-based solid-phase peptide synthesis (SPPS), introducing pseudopeptide bonds during solid-phase. For triazole peptides, the P10 residue was coupled with the azide derivatives of parent amino acids (N3Phe-OH and N3Ala-OH, respectively), followed by copper-catalyzed azide-alkyne cycloaddition (CuAAC) with N-Fmoc-propargylglycine. Nitrogen heterocycles 1b and 2b were obtained by coupling P1 azaGly residue using pre-activated carbonyldiimidazole. All four peptides were purified by RP-HPLC, with purity exceeding 99%.

Eric Biron et al. designed and synthesized a series of peptide nanoscaffolds with two intercalating groups to achieve selective recognition of DNA sequences. The cationic porphyrins were attached to glutamic acid side chains, which were introduced into the peptide sequence using standard solid-phase peptide synthesis methods. The conformation of water-soluble peptide structures carrying two cationic porphyrins was studied using circular dichroism. Using UV-visible spectroscopy and induced circular dichroism, the study demonstrated that these compounds were fully inserted upon binding to double-stranded DNA, exhibiting a strong preference for GC sequences.

(1) Researchers can design and synthesize peptides with specific biological activities, such as mimicking natural hormones or enzymes, for potential therapeutic applications.
(2) Peptides derived from specific regions of proteins can be used to probe interactions with other proteins, thereby gaining deeper insights into cell signaling pathways and protein functions.
(3) Synthetically produced peptides containing immunogenic epitopes can be used to stimulate the immune system and develop vaccines against infectious diseases.
While generally considered low risk, precautions for safe handling are still necessary.
FMOC-Ala-OH is not classified as a hazardous substance. Dust should be avoided, and efforts should be made to minimize dust formation during handling to prevent inhalation.
Wear Gloves: Disposable gloves should be worn to avoid skin contact.
Hand Washing: Thorough hand washing should be done after handling.
Eye Protection: While not always necessary, consider wearing safety goggles as a general precaution for laboratory work.
Cool and Dry: FMOC-Ala-OH should be stored in a cool, well-ventilated place.
Sealed Container: Keep the container tightly sealed to prevent moisture ingress.
Storage Temperature: Long-term storage (beyond 6 months) is recommended below 20°C.
FMOC-Ala-OH, or 9-fluorenylmethoxycarbonyl-L-alanine, is an amino acid derivative widely used in peptide synthesis. Its unique structure makes it an important tool for synthesizing complex proteins and studying protein structure and function. By understanding and applying FMOC-Ala-OH, scientists can more efficiently develop new drugs and explore biochemical mechanisms, providing valuable resources for biomedical research.
[1]https://pubs.acs.org/doi/abs/10.1021/jacs.7b03291
[2]Galibert M, Wartenberg M, Lecaille F, et al. Substrate-derived triazolo-and azapeptides as inhibitors of cathepsins K and S[J]. European Journal of Medicinal Chemistry, 2018, 144: 201-210.
[3]Biron E, Voyer N. Towards sequence selective DNA binding: design, synthesis and DNA binding studies of novel bis-porphyrin peptidic nanostructures[J]. Organic & Biomolecular Chemistry, 2008, 6(14): 2507-2515.
[4]Guangdong Dongyang Pharmaceutical Co., Ltd. Aromatic heterocyclic derivatives and their applications in medicine. 2019-01-18.
[5]https://www.smolecule.com/
[6]https://www.advancedchemtech.com/product/fmoc-ala-oh%E2%88%99h2o/
[7]https://www.abmole.com/literature/fmoc-ala-oh-msds.html
[8]https://www.sigmaaldrich.com/
 |