Glyoxylic acid monohydrate, as a chemical substance, is of significant concern due to glyoxylic acid monohydrate hazards. This article aims to explore the possible dangers it may pose and relevant safety precautions.
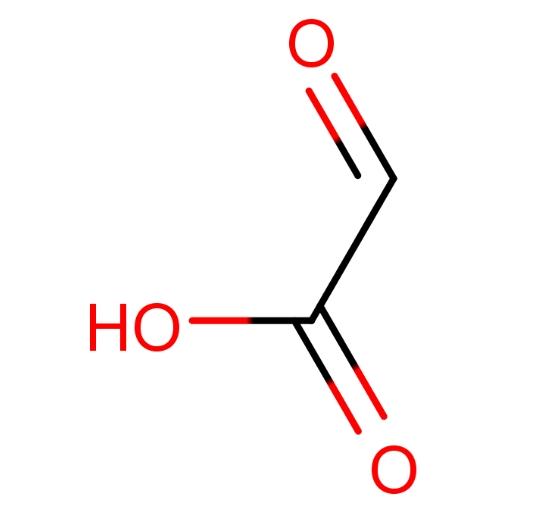
Glyoxylic acid, as an important organic chemical intermediate, finds wide applications in the fields of flavors, medicine, paints, papermaking, and fine chemicals. The primary methods for glyoxylic acid production include glycol nitric acid oxidation, oxalic acid electrolysis, and maleic anhydride ozonolysis. Currently, most domestic glyoxylic acid manufacturers adopt the glycol nitric acid oxidation method, mainly producing 40-50% glyoxylic acid aqueous solutions. Glyoxylic acid monohydrate, as a high-purity glyoxylic acid product, can be used in high-end markets such as fine chemical products. Production of glyoxylic acid monohydrate typically involves maleic anhydride ozonolysis or ozonolysis with a catalyst, processes characterized by advanced technology, high product purity, and minimal environmental impact. The structure of glyoxylic acid monohydrate is as follows:
According to the GHS classification of 29 CFR 1910 (OSHA HCS):
Corrosive to metals (Category 1), H290
Skin irritation (Category 2), H315
Serious eye damage (Category 1), H318
Skin sensitization (Category 1), H317
Skin contact (irritant), eye contact (irritant), ingestion, and inhalation are all highly dangerous. Skin contact (corrosive) and eye contact (corrosive) are hazardous. Skin contact (permeating) poses a slight risk. The extent of tissue damage depends on the duration of contact. Eye contact may lead to corneal damage or blindness. Skin contact can cause inflammation and blistering. Inhaling dust can irritate the gastrointestinal tract or respiratory tract, manifesting as burning, sneezing, and coughing. Severe overexposure can result in lung damage, suffocation, coma, or death. Inflammation of the eyes is characterized by redness, tearing, and itching. Skin inflammation presents as itching, scaling, redness, or occasional blistering.
Skin contact poses a slight hazard (sensitizer). Carcinogenicity: No data available. Mutagenicity: No data available. Teratogenicity: No data available. Developmental toxicity: No data available. The substance may be toxic to mucous membranes, skin, and eyes. Repeated or prolonged exposure to the substance can cause damage to target organs. Repeated eye contact with small amounts of dust can cause eye irritation. Repeated skin contact can lead to local skin damage or dermatitis. Repeated inhalation of dust can cause varying degrees of respiratory tract irritation or lung damage.
Glyoxylic acid releases formaldehyde when heated and is converted to glycine through AGT1 (alanine:glyoxylate aminotransferase 1) or to oxalic acid through glycolate oxidase in peroxisomes in human cells. Formaldehyde is a colorless, toxic aldehyde gas at room temperature.
Show safety data sheet to the attending physician. Seek immediate medical attention.
Immediately flush eyes with plenty of water, also under the eyelids, for at least 15 minutes. Seek immediate medical attention. Keep eye wide open while rinsing.
Immediately wash skin with soap and plenty of water while removing contaminated clothing and shoes. Call a doctor immediately.
Move to fresh air. If breathing stops, perform artificial respiration. Call a doctor or poison control center immediately. Do not give mouth-to-mouth resuscitation if the victim ingested or inhaled the substance; use mouth-to-mouth resuscitation with a pocket mask equipped with a one-way valve or other appropriate respiratory medical device.
Seek immediate medical attention. Do not induce vomiting. Drink plenty of water. Do not give anything by mouth to an unconscious person.
May cause allergic skin reactions. Burns on all contact routes. Product is corrosive material. Do not induce vomiting or lavage. Investigate possible perforation of stomach or esophagus: Ingestion may cause severe swelling, severe tissue damage, and risk of perforation. Symptoms of allergic reactions may include rash, itching, swelling, difficulty breathing, tingling in hands or feet, dizziness, lightheadedness, chest pain, muscle aches, or flushing.
Wear personal protective equipment/face protection. Do not get in eyes, on skin, or on clothing. Use only in a chemical fume hood. Do not breathe dust. Do not eat. If swallowed, seek medical advice immediately.
Keep container tightly sealed and store in a dry, cool, and well-ventilated place. Corrosive area. Keep away from moisture. Store under inert gas. Keep away from incompatible substances: bases, metals, reducing agents, etc.
Wear appropriate protective eyeglasses or chemical safety goggles that comply with 29 CFR 1910.133 or European Standard 166 for OSHA eye and face protection requirements.
Wear appropriate protective gloves and clothing to prevent skin exposure.
Follow OSHA Respiratory Protection Standard 29 CFR 1910.134 or European Standard EN 149. Use respiratory protection approved by NIOSH/MSHA or European Standard EN 149 if exposure limits are exceeded or irritation or other symptoms occur.
In summary, the potential glyoxylic acid monohydrate hazards encompass threats to human health. Therefore, its use and handling should be approached with caution, employing effective protective measures to ensure the safety and health of individuals and the environment.
[1]https://karger.com/cnd/article/12/2/112/827730/Acute-Kidney-Injury-following-Exposure-to
[2]https://www.bio.vu.nl/microb/Protocols/chemicals/
[3]https://pim-resources.coleparmer.com/
[4]https://www.fishersci.com/store/
[5]https://pubchem.ncbi.nlm.nih.gov/
[6]https://www.emdmillipore.com/US/en/product/Glyoxylic-acid-monohydrate,MDA_CHEM-804107
[7]Xinjiang Guolin New Materials Co., Ltd. A crystallization method for glyoxylic acid monohydrate. 2023-10-13.
 |
 |