Hexaflumuron is an effective insect growth regulator widely used in agriculture and sanitation to control pest populations and reduce the risk of disease transmission. Its unique mechanism of action and lasting effects make it an important tool in modern agriculture and disease prevention.
Hexaflumuron, also known as Hexaflumuron, has the chemical name l-[3,5-dichloro-4-(1,1,2,2-tetrafluoroethoxy)phenyl]-3-(2,6-difluorobenzoyl)urea. It belongs to the benzoylurea class of insecticides. Developed by Dow Elanco Company in 1981 and produced in 1987, it was later successfully replicated by Chinese companies such as Shenyang Chemical Research Institute and Dalian Runze (Sha Jia-Jun et al., 1993).

Hexaflumuron is an N-acylurea, where one hydrogen atom on the nitrogen of urea is replaced by a 2,6-difluorobenzoyl group, and the other hydrogen atom is replaced by a 3,5-dichloro-4-(1,1,2,2-tetrafluoroethoxy)phenyl group. It is classified as a benzoylurea insecticide, organic fluorine insecticide, organic chlorine insecticide, dichlorobenzene, N-acylurea, and aromatic ether.
Hexaflumuron, with the chemical formula C16H8Cl2F6N2O3, is a potent insecticide belonging to the N-acylurea class. Its effectiveness relies on a unique combination of structural features, each playing a crucial role in its impact on target pests, especially termites:
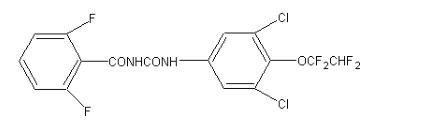
- Core: Central Urea Portion
The foundation of hexaflumuron's structure is the central urea portion, consisting of a carbonyl group connected to two nitrogen atoms. This core unit forms the backbone of the molecule, providing a stable framework for attached functional groups.
- Stability and Activity: 2,6-Difluorobenzoyl Group
One nitrogen atom of the urea part is bonded with a 2,6-difluorobenzoyl group, consisting of a benzene ring with alternating single and double bonds and carbonyl and fluorine atoms at the 2 and 6 positions. The presence of fluorine enhances molecular stability, allowing prolonged environmental persistence. Scientists believe this group contributes to hexaflumuron's bioactivity, possibly affecting its interaction with target molecules in termites.
- Targeting Termites: 3,5-Dichloro-4-(1,1,2,2-Tetrafluoroethoxy)phenyl Group
The other nitrogen atom in the urea part is linked to a more complex group: 3,5-dichloro-4-(1,1,2,2-tetrafluoroethoxy)phenyl. This group includes a benzene ring with chlorine atoms at positions 3 and 5 and a four-carbon chain ending in an ethoxy group. This chain also features four fluorine atoms, highlighting the prevalence of fluorine in the molecular structure. The combination of chlorine and fluorine atoms may help hexaflumuron interact with specific molecules in termites, ultimately disrupting their growth and development.
Hexaflumuron is a colorless solid with a melting point of 202-205°C and a vapor pressure of 0.059 mPa (25°C). Solubility (20°C): 0.027 mg/L in water (18°C), 11.9 mg/L in methanol, and 5.2 g/L in xylene.
- Hexaflumuron is an insect growth regulator (IGR) that inhibits insect growth by interfering with chitin synthesis, essential for termites to form new exoskeletons.
- It is used as part of a pest monitoring system, selectively applied in bait stations where termite activity is present and only used when activity persists. Termites are social insects that share food and foraging locations, recruiting nest mates to feed on hexaflumuron-containing bait.
- Pest control professionals use hexaflumuron as part of above-ground and underground baiting systems, placing it in tamper-resistant bait stations where foraging termites consume it.
Hexaflumuron is a chitin synthesis inhibitor with high insecticidal and ovicidal activity, particularly effective against resistant fourth-generation cotton bollworms. It acts quickly, controlling pests on cotton and fruit trees, including Coleoptera, Diptera, Homoptera, and Lepidoptera, by inhibiting molting and reducing feeding speed, providing a knockdown effect. This product has significant promotional value.
- Persistent Residual Activity
- Effectiveness: Studies show hexaflumuron can be absorbed by termites and transferred within the colony, leading to high mortality rates. However, success in field conditions varies due to termite foraging behavior.
- Limitations: More data is needed to determine how long hexaflumuron remains effective in bait stations under real-world conditions.
- Minimal Environmental Impact
- Effectiveness: Compared to some traditional termite treatments, hexaflumuron generally has lower toxicity.
- Limitations: Long-term environmental impacts require further study.
- Cost-Effectiveness and Return on Investment
- Effectiveness: Opinions vary. Some studies suggest hexaflumuron's concentrated formulations are highly effective with low bait quantities. However, depending on the severity of infestation and bait system efficacy, multiple placements and continuous monitoring may be needed, affecting overall costs.
- Limitations: True cost-effectiveness depends on various factors such as treatment area size, infestation severity, and labor costs associated with installation and monitoring.
Future research on hexaflumuron may explore two main avenues: expanding its application range and reducing its environmental impact. One focus area could be developing hexaflumuron-based products for new pest targets or even human health applications. Additionally, research aims might include creating more targeted formulations that are less persistent in the environment, minimizing potential harm to non-target organisms. Addressing these issues can make hexaflumuron a more versatile and eco-friendly pest control tool.
As an insect growth regulator, hexaflumuron plays a crucial role in agriculture and sanitation. By disrupting pest growth and development, it provides an effective and sustainable method for controlling pest populations and reducing disease transmission risks. Although its application requires caution, continued advancements in science and technology will ensure hexaflumuron remains vital for crop protection, agricultural productivity, and public health.
[1]http://npic.orst.edu/factsheets/archive/
[2]https://pubchem.ncbi.nlm.nih.gov/compound/91741
[3]https://en.wikipedia.org/wiki/Acylurea
[4]Xue Beibei. Preparation and stability study of hexaflumuron aqueous suspension [D]. Shandong Agricultural University, 2011.
[5]https://link.springer.com/article/10.1007/s11356-013-2036-8
 |
 |
 |