This article will discuss the degradation of Methyl 2,4-dihydroxybenzoate (MDB) under visible light irradiation, aiming to provide reference ideas and basis for researchers in related fields.
Background:
Methyl 2,4-dihydroxybenzoate (MDB) is a hydroxylated product of methyl salicylate in the photocatalytic degradation process, more stable than the methyl ester, and with stronger acute toxicity. As a pharmaceutical intermediate, MDB has irritant effects on the eyes, respiratory system, and skin, posing potential risks to water resources and ecosystems. Therefore, the removal of MDB is crucial.
Degradation:
Lu Mingli et al. proposed the catalytic degradation of MDB using microwave-assisted synthesized iodine-doped Bi4O5Br2 (I-Bi4O5Br2) under visible light irradiation and achieved good progress. The specific experimental process is as follows:
(1) Synthesis of Bi4O5Br2 and I-Bi4O5Br2 photocatalysts
Using bismuth nitrate pentahydrate as the bismuth source, potassium bromide and potassium iodide as bromine and iodine sources, and ethylene glycol (EG) as the solvent, the synthesis was carried out using the microwave method. Specifically, 0.485 g of Bi(NO3)3·5H2O was added to 50 mL of EG and stirred magnetically until completely dissolved. Then, 0.5 mmol of KBr was added to the solution, followed by the addition of 0.35 mmol of KI after complete dissolution. The solution was transferred to a 150 mL round-bottom flask and reacted under microwave power of 400 W for 4 minutes. After the reaction, the mixture was cooled to room temperature. The precipitate was collected by centrifugation, washed with deionized water and ethanol several times, and dried at 60°C in an oven for 12 hours to obtain the I-Bi4O5Br2 photocatalyst. In the preparation of the reference sample Bi4O5Br2 photocatalyst, the addition of KI was omitted, and other steps remained the same.
(2) Photocatalytic performance testing
The photocatalytic degradation experiment was conducted using a photochemical reaction instrument (XPA-VII) with a 1000 W xenon lamp as the visible light source. A 420 nm filter was used to filter out ultraviolet light, and the entire reaction system was equipped with circulating cooling water to maintain the reaction at room temperature. At the beginning of the reaction, 50 mg of catalyst was added to 50 mL of MDB solution with a mass concentration of 20 mg/L. Before turning on the light, the suspension was stirred in the dark for 60 minutes to ensure adsorption-desorption equilibrium between the catalyst and the pollutant. After turning on the light, 2.5 mL of solution was taken at regular intervals, filtered through a 0.45μm aqueous membrane filter to remove solids, and then the absorbance was measured using a UV-visible spectrophotometer (UV-1800, Shimadzu) to analyze the degradation rate, and the degradation rate was used to measure the catalytic degradation activity of the catalyst.
(3) Results
Compared with pure Bi4O5Br2, the I-Bi4O5Br2 catalyst exhibited excellent visible light degradation activity towards MDB, with a degradation rate five times that of pure Bi4O5Br2 (0.038 min^-1). The main reason for the enhanced photocatalytic activity of I-Bi4O5Br2 is its better separation efficiency of photogenerated electron-hole pairs and broader light absorption range. Mechanistic studies indicate that photogenerated holes and superoxide radicals are the main active oxygen species in the visible light photocatalytic degradation process of MDB.
Applications:
Fluorescein was first synthesized by Von Bayer in 1871 through the condensation of resorcinol and phthalic anhydride in the presence of zinc chloride. Its maximum absorption and emission wavelengths in aqueous solution are 492 nm and 517 nm, respectively, with a quantum yield of up to 0.92 at pH > 8. Due to its high fluorescence intensity, many fluorescein derivatives have been synthesized and used as fluorescent probes, such as 5(6)-carboxyfluorescein and 5(6)-carboxyfluorescein succinimidyl ester, which are the most widely used fluorescent derivatization reagents.
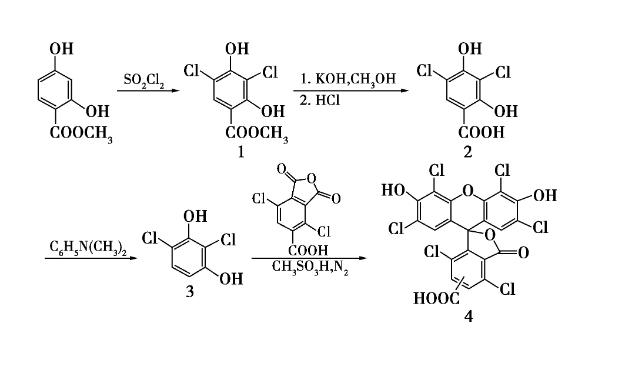
Using Methyl 2,4-dihydroxybenzoate as the raw material, 2,4-dichlororesorcinol was synthesized through chlorination, hydrolysis, and decarboxylation reactions. Under the action of methylsulfonyl chloride, important chlorinated fluoresceins were obtained by condensing 2,4-dichlororesorcinol with 3,6-dichloro-4-carboxyphthalic anhydride. The synthesis route is as follows:
References:
[1] Lu Mingli, Xiao Xin, Zhang Ling, et al. Visible light photocatalytic degradation of Methyl 2,4-dihydroxybenzoate using iodine-doped Bi_4O_5Br_2 [J]. Journal of South China Normal University (Natural Science Edition), 2019, 51(01): 22-27.
[2] Wu Xianglong, Tian Min, Tong Meng, et al. Synthesis of chlorinated fluoresceins [J]. Chemical Reagents, 2008, (10): 771-772+778. DOI:10.13822/j.cnki.hxsj.2008.10.011.