Brief Overview:
Dibucaine Hydrochloride, chemical name: 2-butoxy-N-(2-diethylaminoethyl)-quinoline-4-carboxamide hydrochloride, is used for epidural anesthesia, spinal anesthesia, with local anesthetic potency 22-25 times greater than procaine, and longer duration. It appears as a white or nearly white crystalline powder, odorless and tasteless, slightly hygroscopic. It is highly soluble in water, methanol, ethanol, and glacial acetic acid, soluble in acetone, dimethyl sulfoxide or chloroform, insoluble in ether. Melting point is 95-97.5℃.
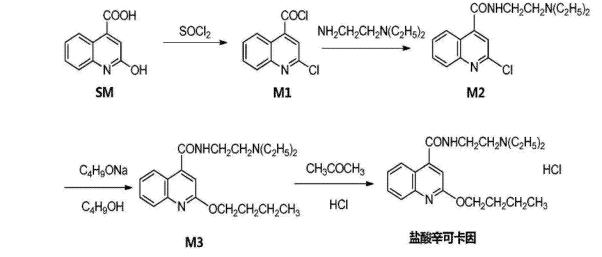
Synthesis:
(1) Synthesis of 2-Chloro-4-quinolinecarboxylic Acid Chloride
2-Hydroxy-4-quinolinecarboxylic acid (15-25 g) was added to a 250 mL three-neck flask at room temperature with stirring, followed by dropwise addition of thionyl chloride (12-18 g) in toluene (120-180 mL), warmed to 50-70℃, and reacted for 2.5-3.5 hours. After cooling to 25-35℃, it was concentrated under reduced pressure, then a small amount of toluene was added, and continued concentration to dryness to obtain 2-chloro-4-quinolinecarboxylic acid chloride.
(2) Synthesis of 2-Chloro-N-[2-(diethylamino)ethyl]-4-quinolinecarboxamide
The obtained 2-chloro-4-quinolinecarboxylic acid chloride was diluted with toluene (200-300 mL) and added to a 500 mL three-neck flask, followed by addition of N,N-diethylethylenediamine (6-12 g). The mixture was warmed to 60-80℃, stirred until complete reaction, cooled to room temperature, then water was added and stirred for 25-35 minutes. After separation, the organic layer was washed with water once to three times, then with saturated brine one to two times, dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure to obtain 2-chloro-N-[2-(diethylamino)ethyl]-4-quinolinecarboxamide.
(3) Synthesis of Dibucaine
Butanol (120-180 mL) was added to a 500 mL reaction flask, nitrogen-protected, and 2.5-4 g of metallic sodium was added. After stirring at room temperature, the obtained 2-chloro-N-[2-(diethylamino)ethyl]-4-quinolinecarboxamide was added portionwise, gradually heated to reflux, and maintained for 4-8 hours. After cooling to room temperature, purified water (50-150 mL) was added, stirred for 30 minutes, allowed to settle, then the aqueous layer was discarded after 30 minutes. The organic layer was dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure. Then, 20-40 mL of water and 100-200 mL of n-hexane were added, stirred and heated to 40-60℃, allowed to settle for 10-20 minutes, separated, and water layer was added to the organic layer, stirred and heated to 40-60℃, allowed to settle for 10-20 minutes, the water layer was removed. The n-hexane layer was collected and allowed to crystallize overnight in a freezer to obtain crude dibucaine; then, 10-20 g of crude dibucaine was added to a 100 mL three-neck flask with 30-50 mL of ethyl acetate, dissolved by heating, kept warm at 40-60℃ for 20-40 minutes, filtered while hot, the filtrate was placed in the refrigerator for crystallization, filtered, and dried to obtain purified dibucaine.
(4) Synthesis of Dibucaine Hydrochloride
To a 300 mL three-neck flask, 12-20 g of purified dibucaine, 15-30 mL of acetone were added, and slowly heated to 40-70℃. After complete dissolution, hydrochloric acid in acetone solution was added dropwise, with a mass ratio of hydrochloric acid to acetone of 1:0.5-2. After completion of the dropwise addition, the mixture was kept warm and stirred for 0.5-1 hour, filtered while hot, the filtrate was crystallized in the refrigerator, filtered, and dried to obtain dibucaine hydrochloride.
Detection:
Huang et al. established an HPLC method for determining the content of dibucaine hydrochloride in compound polyoxyethylene sorbitan monolaurate cream. The chromatographic column was Agilent TC-C18 (250mm×4.6mm, 5μm); the mobile phase was acetonitrile-0.02mol·L-1 potassium dihydrogen phosphate solution (adjusted to pH 4.6 with 0.1mol·L-1 potassium hydroxide solution) (40:60); detection wavelength was 230nm, and the flow rate was 1.0mL·min-1. The results showed that dibucaine hydrochloride had a good linear relationship within the detection range of 2.1333μg·mL-1 to 21.3332μg·mL-1 (r=0.99996, n=6), with an average recovery rate of 100.50% (n=9, RSD=0.8%). This method is simple to operate, highly sensitive, and reproducible.
References:
[1] Beijing Lianben Pharmaceutical Chemistry Technology Co., Ltd., Beijing Jikanglitong Pharmaceutical Technology Co., Ltd. A method for preparing high purity dibucaine hydrochloride. 2012-08-15.
[2] Kunming Yuanrui Pharmaceutical Co., Ltd. A method for preparing dibucaine hydrochloride. 2017-03-15.
[3] Huang L, Huang J. Determination of dibucaine hydrochloride in compound polyoxyethylene sorbitan monolaurate cream by HPLC. Strait Pharmaceutical Journal. 2008;(08):48-49.