Understanding the hydroxypropyl beta cyclodextrin toxicity and corresponding safety measures is crucial to ensuring its safe application in fields such as pharmaceuticals and food industries. Comprehensive knowledge of its toxicological characteristics and safe usage methods helps minimize potential risks and ensures its effective functionality.
Hydroxypropyl beta cyclodextrin (HP-β-CD), also known as 2-hydroxypropyl-β-cyclodextrin, possesses a unique stereochemical structure with outer hydrophilic and inner hydrophobic properties typical of cyclodextrin compounds. Compared to natural β-cyclodextrin, HP-β-CD exhibits higher water solubility and bioavailability, making it more effective in enhancing the solubility of poorly soluble substances in water and improving environmental stability. Hence, HP-β-CD has gained widespread attention and application in biomedical, food, analytical chemistry, and environmental separation fields.
HP-β-CD is a hydroxyalkyl derivative and is generally considered relatively non-toxic in toxicological studies.
According to Sarah Gould et al., HP-β-CD demonstrates good tolerance in tested animal species (rats, mice, and dogs), particularly when administered orally, with limited toxicity. Short-term studies indicated slight biochemical changes, while longer studies up to three months showed additional minor hematological changes but no histopathological alterations. Intravenous injection resulted in histopathological changes in the lungs, liver, and kidneys, all of which were reversible and did not reach significant levels. Carcinogenicity studies in rats indicated increased pancreatic and intestinal tumors, both considered rat-specific. Non-carcinogenic changes were also observed in the urinary tract, but these were reversible and did not impair kidney function. No adverse effects on embryo-fetal development were observed in either rats or rabbits. HP-β-CD has demonstrated good tolerability in humans, with the main adverse event being diarrhea, and no adverse events related to kidney function have been recorded to date.
Sarah Gould et al. conducted a series of toxicity studies on HP-β-CD involving mice, rats, monkeys, and dogs, with study durations up to 12 months. Additional studies by AstraZeneca (AZ) on rats and dogs lasted up to one month. Table 1 summarizes AZ's data.
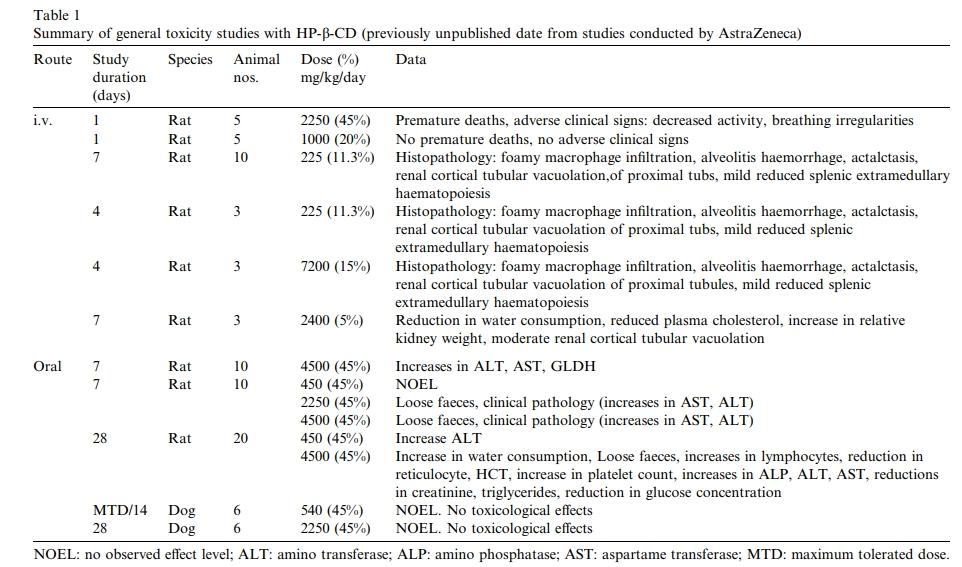
In mice, acute injection of HP-β-CD at doses as high as 10,000 mg/kg bw intraperitoneally was neither lethal nor produced any toxicity (Fromming and Szejtli, 1996).
In crab-eating macaques, a single intravenous injection of 50% w/v HP-β-CD at 10,000 mg/kg did not cause death (Brewster et al., 1990). In mice, a single intravenous injection of up to 2000 mg/kg bw also did not result in lethality (Fromming and Szejtli, 1996).
Rats were continuously infused via the femoral vein with 225 mg/kg/day of 11.25% w/v HP-β-CD for 4-7 days. Histopathological changes included alveolar macrophage infiltration with pulmonary inflammation hemorrhage and atelectasis. Additionally, mild vacuolization of the proximal convoluted tubules in the renal cortex and slight reduction in extramedullary hematopoiesis in the spleen were observed with administration of 15% HP-β-CD and 2.5% glucose for 4 days in rats.
Details from existing literature are limited. There is no evidence of genotoxicity of HP-β-CD in Ames tests (up to 1000 μg/plate) and in vivo micronucleus tests (up to 5000 mg/kg/day; species unknown) (Coussement et al., 1990). HP-β-CD has also shown negative results in unscheduled DNA synthesis (UDS) assays (for DNA damage), mouse lymphoma tests (for gene mutation), and human lymphocyte tests (for chromosomal aberrations).
Literature reports include developmental hydroxypropyl beta cyclodextrin toxicity studies in rats and rabbits via oral or intravenous administration. In embryo-fetal development studies in rats (administered from gestation days 6-16) via intravenous injection, 400 mg/kg/day caused slight maternal toxicity with no adverse effects observed in offspring. Similar studies in rabbits (from gestation days 6-18) did not show adverse effects at doses up to 400 mg/kg/day (Coussement et al., 1990). Oral studies in rats for teratogenic and embryo toxicity (administered from gestation days 6-16) at doses up to 400 mg/kg/day showed no maternal toxicity, embryo toxicity, or teratogenicity. Oral studies in rabbits (from gestation days 6-18) at 1000 mg/kg revealed slight maternal and embryo toxicity (Coussement et al., 1990).
Clinical studies have reported good tolerance in most patients treated with HP-β-CD, with doses ranging from 4-8 grams per day for at least 2 weeks (Irie and Uekama, 1997). Increased occurrence of loose stools and diarrhea was noted in volunteers receiving higher oral doses of 16-24 grams per day continuously for 14 days. Therefore, based on these clinical data, HP-β-CD is considered non-toxic if daily doses are below 16 grams (for at least 14 days). In a study involving intravenous administration (Seiller et al., 1990), a single dose of up to 3 grams did not show any measurable impact on kidney function, and all volunteers exhibited good tolerance. No adverse reactions were reported following a one-week study of a single intravenous dose of 1 gram (Janssen Technical Bulletin, 1992).
Cyclodextrins, a class of cyclic oligosaccharides, feature a hydrophilic outer surface and a hydrophobic internal cavity. Apart from other uses, cyclodextrins (especially β-cyclodextrins) are widely used as pharmaceutical excipients because they can enhance the solubility and dissolution rate of poorly soluble drugs. 2-Hydroxypropyl-β-cyclodextrin consists of seven α-(1,4)-linked glucopyranose units with randomly substituted hydroxypropyl groups on the β-cyclodextrin molecule. Johnson & Johnson was the first to use 2-hydroxypropyl-β-cyclodextrin in drug formulations, with its marketed product Sporanox? (itraconazole) currently found in over 35 marketed drugs. β-Cyclodextrin is listed in the FDA's Generally Recognized as Safe (GRAS) list, and its derivative HP-β-CD is included in USP/NF and EP and is listed as an inactive ingredient by the FDA. Extensive data in the literature demonstrates good tolerance of HP-β-CD in most species, with doses up to 470 mg/kg/day (30 g within 4 days) in human volunteers and a single dose of up to 3.0 grams. However, HP-β-CD is considered only as a complexing agent and has not yet received FDA approval as a therapeutic drug product.
Regulatory approvals come with specific guidance. The permissible limits for HPBCD use are clearly defined to ensure safe incorporation into medications. These limits are in place to mitigate any potential adverse effects associated with HPBCD and prioritize patient safety. The US Food and Drug Administration (FDA) categorizes 2-hydroxypropyl-β-cyclodextrin and 2-hydroxypropyl-γ-cyclodextrin (HPγCD) as approved inactive ingredients, suitable for oral and intravenous administration for HP-β-CD, while HPγCD can only be used in topical products at a maximum concentration of 1.5% (w/v) (FDA, 2016).
Understanding the hydroxypropyl beta cyclodextrin toxicity and implementing appropriate safety measures is crucial for its safe application across various fields. Through scientific assessment of its toxicological characteristics and effective safety measures, potential health risks can be minimized, ensuring its beneficial role in pharmaceuticals, food industries, and beyond, thereby providing greater assurance for human health and quality of life.
[1] Fan B, Wei G, Yu Y, et al. Study on Preparation of Hydroxypropyl-β-Cyclodextrin by Normal Pressure Dehydration. Journal of Beijing University of Chemical Technology (Natural Science Edition). 2014;41(05):53-58. DOI:10.13543/j.cnki.bhxbzr.2014.05.005.
[2] https://www.researchgate.net/publication/21092840_Use_of_2-Hydroxypropyl-b-cyclodextrin_as_a_Solubilizing_and_Stabilizing_Excipient_for_Protein_Drugs
[3] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6995511/
[4] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4048128/
[5] https://www.sciencedirect.com/science/article/abs/pii/S0278691505001006
[6] https://www.zyversa.com/pipeline/about-hydroxypropyl-beta-cyclodextrin
 |