Imazethapyr, also known as Pursuit, Pursuit, and Imazethapyr, chemical name 5-ethyl-(4-isopropyl-4-methyl-5-oxo-2-imidazolin-2-yl)nicotinic acid, is an imidazolinone herbicide developed successfully by the former American Cyanamid Company (now BASF) in 1984. It is highly efficient and broad-spectrum, low-toxicity herbicide used in soybean fields, effective against over 20 types of monocotyledonous and dicotyledonous weeds, particularly suitable for soybean fields [1]. There are many synthesis methods for imazethapyr, such as ethyl chloroacetate esterification method, 5-ethylpyridine-2-carboxylic acid method, and 5-ethyl-2-pyridine method.
Imidazolinone herbicides inhibit acetolactate synthase (ALS) or acetohydroxyacid synthase (AHAS), absorbed by roots, stems, and leaves of plants, transmitted to the xylem and phloem, accumulating in meristematic tissues, and inhibiting the biosynthesis of branched-chain amino acids—valine, leucine, and isoleucine, thereby inhibiting the acetolactate synthase in plants, disrupting protein synthesis, interfering with DNA synthesis and cell division. After stem and leaf treatment, sensitive weeds immediately cease growth, with death occurring in about 2-4 weeks; weeds treated in soil have necrosis of apical meristems, cease growth, though some weeds can germinate, when plants grow to 2-5 cm, growth will cease, and death will follow.
Imazethapyr is a post-emergence herbicide that, when mixed, is rapidly absorbed by plant leaves and transferred throughout the entire plant. Weeds stop growing shortly thereafter and die in about 4-6 weeks. Like other imidazolinone herbicides, it primarily inhibits the activity of acetohydroxyacid synthase, causing disruption to the biosynthesis of three branched-chain amino acids—valine, leucine, and isoleucine—thus interfering with protein and DNA synthesis and cell division.
Inhibition of acetolactate synthase (ALS), also known as acetohydroxyacid synthase (AHAS), is a key enzyme in the biosynthesis of branched-chain amino acids isoleucine, leucine, and valine.
Medium to long (4-6 months), depending on weather and soil conditions (more persistent under dry conditions).
Imazethapyr is a systemic herbicide absorbed by roots and leaves, transferred to the xylem and phloem, and accumulated in meristematic tissues. It controls major annual and perennial grasses and broadleaf weeds in soybeans, peanuts, and other legume crops. It inhibits amino acid synthesis, disrupting protein and DNA synthesis. Imazethapyr is classified as "Toxicity Category III" by WHO (a.i.) and EPA (formulation).
Three years after the sulfonylurea herbicide was introduced, American Cyanamid Company successfully developed another type of highly active, broad-spectrum, low-toxicity herbicide—imidazolinone herbicide—and subsequently developed excellent varieties such as imazethapyr, imazapyr, methoxyimazethapyr, imazaquin, and imazethapyr (Imazethapyr). This type of herbicide is known for its broad spectrum of weed control, effective against annual and perennial monocotyledonous and dicotyledonous weeds and monocots. American Cyanamid Company is seeking new use registrations for two previously registered herbicides: imazethapyr is currently registered for peanuts, soybeans, and other legumes, while imazapyr is currently registered for non-agricultural land areas and forests. Below are detailed descriptions of both:
Imazapyr, 2-(4-isopropyl-4-methyl-5-oxo-2-imidazolin-2-yl)nicotinic acid, is a non-selective herbicide for non-crop land applications, particularly effective against difficult-to-control perennial grasses. Its mode of action is non-selective, absorbed by leaves and rapidly translocated. Imazapyr is absorbed by leaves and roots and quickly moves within the plant. It accumulates in the meristematic tissue region (active growth area) of plants. In plants, imazapyr disrupts protein synthesis and interferes with cell growth and DNA synthesis. It controls vegetation by disrupting enzyme pathways. Inhibition of acetolactate synthase. It is used to control a wide range of weeds, including terrestrial annual and perennial grasses and broadleaf herbaceous plants, woody species, riparian and emergent aquatic species. It is used to eliminate dense stands of tan oak (Tan Oak) and Pacific madrone (Pacific Madrone). In addition, imazapyr is also used to control annual and perennial weeds, broadleaf weeds, shrubs, vines, and many deciduous trees.
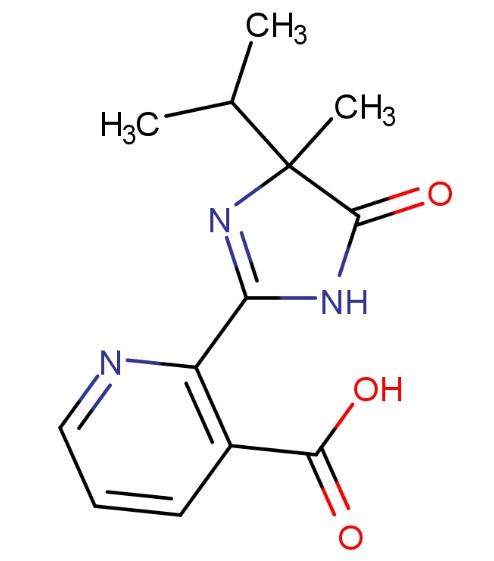
Imazethapyr, 5-ethyl-2-[(RS)-4-isopropyl-4-methyl-5-oxo-2-imidazolin-2-yl]nicotinic acid, belongs to the imidazolinone family, toxicity level III, and is used as a selective herbicide for controlling a variety of weeds. Its excellent weed control effectiveness in legumes and other legume crops and good crop tolerance contribute to its popularity. Imazethapyr lowers levels of three branched-chain fatty amino acids (isoleucine, leucine, and valine) by inhibiting acetohydroxyacid synthase (AHAS), which is a common enzyme in the biosynthesis pathway of these amino acids. This inhibition disrupts protein synthesis, leading to interference with DNA synthesis and cell growth. Under field conditions, imazethapyr dissipates in soil through microbial degradation and photolysis.
Imazethapyr achieves excellent weed control by disrupting specific physiological processes in plants, such as cell division and lipid synthesis pathways. This mode of action not only significantly impacts weeds but also provides an effective alternative to glyphosate in agricultural production. However, to ensure optimal effectiveness and safety, users should strictly follow label instructions while being mindful of potential impacts on the environment and non-target plants.
[1] https://cals.cornell.edu/weed-science/herbicides/herbicide-reference/pursuit-imazethapyr
[2] https://www.indiamart.com/proddetail/imazathapyr-systemic-herbicide-22266740333.html
[3] https://www.jstage.jst.go.jp/article/jpestics/40/3/40_D14-109/_html/-char/en
[4] https://trid.trb.org/View/302115
[5] Yu Fei, Li Ziliang, Lin Yang, et al. Synthesis of 5-ethyl-2,3-pyridinedimethanol diethyl ester intermediate of imazethapyr [J]. Pesticides, 2019, 58 (12): 875-877+887. DOI: 10.16820/j.cnki.1006-0413.2019.12.004.
[6] Liu Liping. Research on the synthesis process of herbicide imazethapyr [D]. Heilongjiang University, 2012.
[7] https://pubchem.ncbi.nlm.nih.gov/
 |
 |