Welcome to the world of molecular structures! Today, we'll explore the Lewis structure of propane (C3H8), a simple yet essential hydrocarbon widely used as a fuel and chemical feedstock. Understanding its Lewis structure helps us grasp its bonding and molecular properties.

What is the Lewis Structures?
Lewis structures, pioneered by Gilbert N. Lewis, are diagrams that represent the arrangement of valence electrons in a molecule. By depicting atoms as symbols and showing valence electrons as dots or lines, Lewis structures reveal the connectivity of atoms and help predict molecular geometry and properties based on electron pair arrangements.
What is Propane?
Propane (C3H8) is a three-carbon alkane belonging to the family of saturated hydrocarbons. It is a colorless, odorless gas at standard temperature and pressure but can be liquefied under modest pressure for easy storage and transportation. Propane is a crucial energy source used for heating, cooking, and as fuel for vehicles and industrial processes.
How to draw Lewis structures for Propane?
Let's outline the steps to draw the Lewis structure of propane:
Step 1: Count Total Valence Electrons: Carbon contributes 3 valence electrons each, and hydrogen contributes 1, giving a total of 3 + (8 x 1) = 11 valence electrons for propane.
Step 2: Connect Atoms with Single Bonds: Arrange the carbon atoms in a chain, each bonded to three hydrogen atoms. Distribute remaining electrons as lone pairs to satisfy the octet rule.
Step 3: Verify Octet Rule: Ensure each carbon atom has 8 electrons (including bonding and lone pairs) and each hydrogen atom has 2 electrons (a full valence shell).
Step 4: Finalize Structure: The Lewis structure of propane consists of a chain of three carbon atoms bonded to eight hydrogen atoms.

Molecular geometry of Propane
The molecular geometry of propane can be described as tetrahedral around each carbon atom. Each carbon is bonded to four other atoms, resulting in a tetrahedral arrangement with bond angles of approximately 109.5 degrees.
Hybridization in Propane
In propane, each carbon atom undergoes sp3 hybridization. One s orbital and three p orbitals combine to form four sp3 hybrid orbitals. These orbitals then overlap with the 1s orbital of hydrogen atoms to form four strong σ bonds, resulting in a tetrahedral geometry.
Propane (C3H8) is a nonpolar molecule. While carbon-hydrogen bonds are slightly polar due to the electronegativity difference, the symmetrical arrangement of carbon and hydrogen atoms in propane cancels out any overall dipole moment, resulting in a nonpolar molecule.
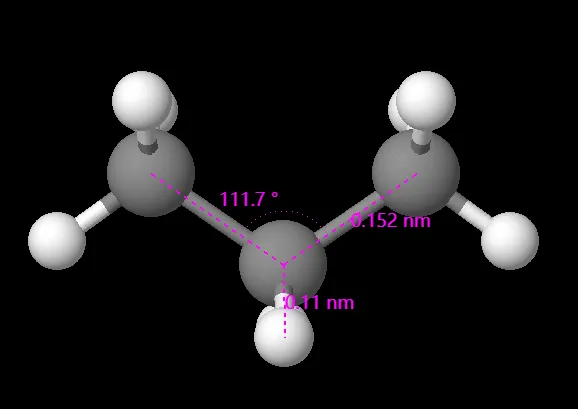
What are approximate bond angles and Bond length in Propane?
The bond angle in propane is approximately 109.5 degrees, consistent with tetrahedral geometry around each carbon atom. The carbon-carbon bond length in propane is approximately 154 picometers (pm), while the carbon-hydrogen bond length is around 109 pm.
Note: Actual bond angles and lengths may slightly vary due to factors such as steric effects and bond rotation.
Highlight of Propane
| Propane Cas 74-98-6 |
| Molecular formula |
C3H8 |
| Molecular shape |
- |
| Polarity |
nonpolar |
| Hybridization |
sp3 hybridization |
| Bond Angle |
109.5 degrees |
| Bond length |
154 pm |





