Piperidine is an important six-membered nitrogen heterocyclic compound, and many natural products contain the structural unit of piperidine. Piperidine derivatives have significant research value in the pharmaceutical field, widely used in the synthesis of analgesics, anti-inflammatory agents, antihistamines, antiarrhythmic drugs, antipsychotics, and antitumor drugs. Introducing the piperidine structural unit into drug molecules has become an essential means of drug development.
N-Benzyl-4-piperidone, with the molecular formula C12H15NO, is an intermediate in many drugs, widely used in the synthesis of analgesics, antiviral agents, antipsychotics, and antitumor drugs.
Ye Hong et al. used benzylamine and methyl acrylate as starting materials, employing microwave radiation for Michael addition and Dieckmann condensation. They tested different microwave radiation times, powers, and temperatures. The optimal Michael addition conditions were set at a microwave power of 110-120 W, with a microwave reaction temperature of 50-55°C and a radiation time of 80 minutes. The best Dieckmann condensation conditions were set at a microwave power of 90-100 W, with a microwave reaction temperature of 70-75°C and a radiation time of 20 minutes. The final product, N-Benzyl-4-piperidone, was synthesized through decarboxylation by heating and hydrolysis.
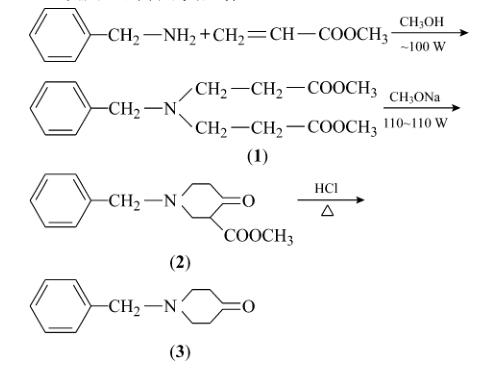
Zhao Aiming et al. reported a method for preparing N-Benzyl-4-piperidone, including the following steps:
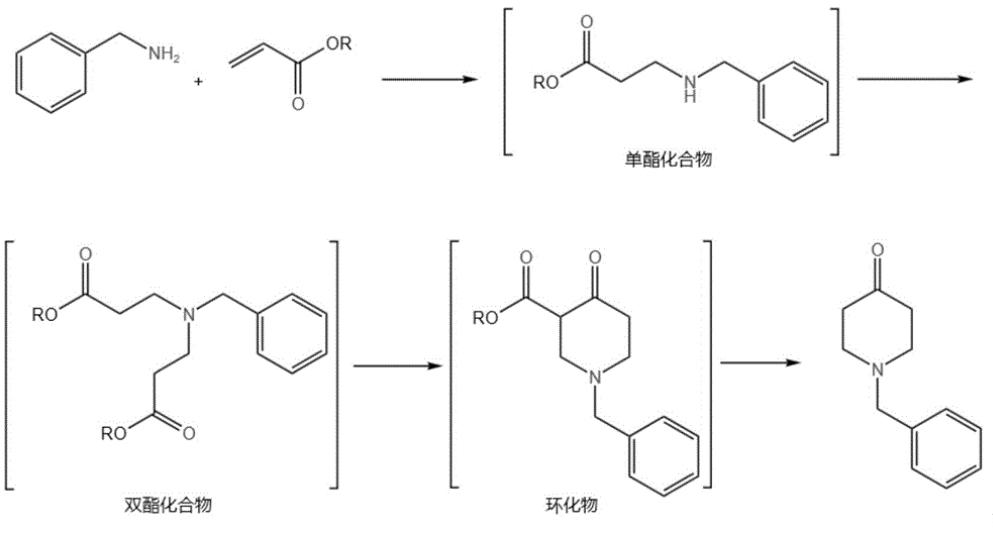
Add benzylamine and an alcohol-based organic solvent to a reactor, and then gradually add acrylate. The molar ratio of acrylate to benzylamine is 2.6-5. Stir the mixture for about an hour, then raise the temperature to 50-60°C and maintain it for 9–24 hours. Excess acrylate helps reduce the amount of monoester compounds and increase the purity of the product. After the reaction, recover the excess acrylate and alcohol-based organic solvent by distillation under normal or reduced pressure, preferably under normal pressure to increase the recovery rate.
Add a condensation reaction organic solvent, and add an organic base in batches. The organic base can be selected from sodium methoxide, sodium ethoxide, or potassium tert-butoxide. Heat to 50-85°C and maintain the reaction for 9–16 hours, preferably 12 hours at 80°C. Low-boiling substances are evaporated during the reaction to maintain the temperature.
After the second step, cool the mixture, neutralize it with acid, then add a catalyst and maintain the reaction at 60-85°C for 1–5 hours. The catalyst can be selected from lithium chloride or calcium chloride, with a molar ratio of catalyst to benzylamine of 0.05-0.5.
The benzyl group of N-Benzyl-4-piperidone can be hydrogenated and removed, allowing different groups to be introduced through alkylation, acylation, and other reactions. The carbonyl group at the 4-position is also a crucial reactive site, with high activity, and can easily be converted into hydroxyl, halogen, amino, cyano, carboxyl, ester, formyl, and other active groups under certain conditions. This makes it an important intermediate in the synthesis of piperidine-based drugs. For example:
Synthesis of Donepezil: N-Benzyl-4-piperidone is a key intermediate in Donepezil. Donepezil is a second-generation, specific, reversible central acetylcholinesterase (AChE) inhibitor with minimal effects on peripheral AChE. Its primary mechanism is to inhibit AChE activity, slowing the degradation of acetylcholine (ACh) in the synaptic cleft, thereby increasing ACh concentration and improving cognitive function in Alzheimer's disease (AD) patients.
Synthesis of Alfentanil and Sufentanil: N-Benzyl-4-piperidone is the starting material for synthesizing fentanyl. Shi Yubai et al. synthesized 1-Benzyl-4-phenylaminocyanopiperidine by reacting N-Benzyl-4-piperidone with aniline and trimethylsilyl cyanide. After hydrolysis with concentrated sulfuric acid and potassium hydroxide, esterification, reduction, and methylation, 1-Benzyl-4-phenylaminomethoxymethylpiperidine was obtained, which was further processed to synthesize the intermediates N-(4-methoxymethylpiperidine-4-yl)-N-phenylpropionamide hydrochloride for alfentanil and sufentanil, with a total yield of about 25% (based on N-Benzyl-4-piperidone).
In organic synthesis research, N-Benzyl-4-piperidone is a key reagent for synthesizing complex organic molecules due to its diverse reaction characteristics. For example:
Sun Jufeng et al. used N-Benzyl-4-piperidone and benzaldehyde derivatives as raw materials to synthesize five N-Benzyl-3,5-di(arylmethylene)-4-piperidone derivatives through an aldol condensation reaction catalyzed by dry HCl gas at 20°C for 7–8 hours. The reaction progress was monitored by TLC, and the target compounds were obtained through recrystallization. The resulting compounds exhibited good inhibitory effects on several tumor cells and represented a novel class of cytotoxic agents with multidrug resistance reversal activity.
Liu Zhanpeng et al. synthesized the pharmaceutical intermediate 4-(3-trifluoromethyl)phenyl-4-piperidinol with a total yield of 87.9% and a purity of 99.0% using m-bromotrifluoromethylbenzene and N-Benzyl-4-piperidone as raw materials through a Grignard reaction and catalytic debenzylation.
 |