
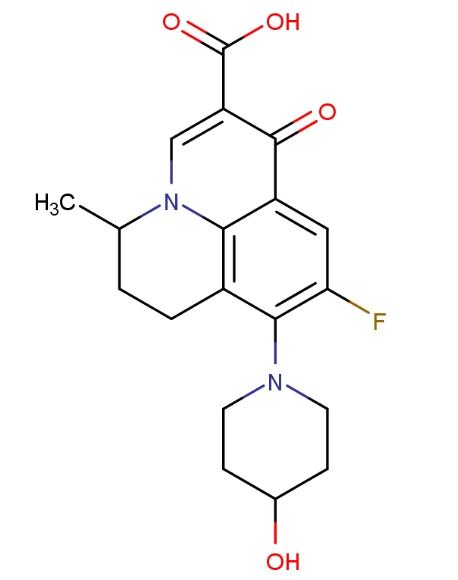
Nadifloxacin, chemically known as (±)-9-fluoro-6,7-dihydro-8-(4-hydroxy-1-piperidinyl)-5-methyl-1-oxo-1H,5H-benzo[i,j]quinolizine-2-carboxylic acid, is the world's first topical quinolone antibiotic with a benzoquinoline skeleton, developed by Otsuka Pharmaceutical Co., Ltd. in Japan. It is mainly used to treat acne and was first marketed in Japan in 1993 as a 1% ointment. Its efficacy surpasses traditional antibiotics such as tetracycline, erythromycin, and clindamycin. The structure of nafloxacin is as follows:
Nadifloxacin is effective against Gram-positive, Gram-negative, and anaerobic bacteria. Studies have shown that nearly 80% of individuals aged 12 to 25 suffer from acne, and nadifloxacin has significant antibacterial effects against clinical isolates such as Propionibacterium acnes and coagulase-negative staphylococci, with minimal resistance development. This potential market makes the development of safe and effective acne treatments highly promising.
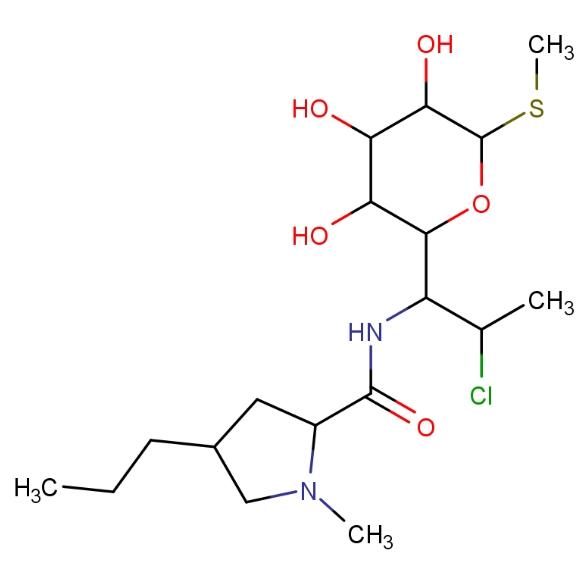
Clindamycin is a lincosamide antibiotic used to treat a variety of bacterial infections, including osteomyelitis (bone infections), joint infections, pelvic inflammatory disease, streptococcal pharyngitis, pneumonia, acute otitis media (middle ear infections), and endocarditis. It is also used to treat acne and certain methicillin-resistant Staphylococcus aureus (MRSA) infections. Clindamycin can be administered orally, intravenously, or topically as a cream or gel for skin or vaginal application. The structure of clindamycin is as follows:
Nadifloxacin inhibits the enzyme DNA gyrase, which is involved in bacterial DNA synthesis and replication, thereby inhibiting bacterial proliferation. In addition to its therapeutic antibacterial effect, nadifloxacin also has sebum-suppressing and anti-inflammatory effects, which help improve the clinical condition of patients.
Clindamycin primarily has a bacteriostatic effect, and at higher concentrations, it may have a bactericidal effect. It inhibits bacterial protein synthesis by interfering with ribosomal translocation, similar to macrolides. Clindamycin binds to the 50S ribosomal subunit of bacteria, overlapping with the binding sites of oxazolidinones, pleuromutilins, and macrolide antibiotics. This binding is reversible, and clindamycin is more effective than lincomycin.
In a study by Sanjay Kumar and colleagues, the efficacy of nadifloxacin vs clindamycin in patients with acne vulgaris was compared in terms of inflammatory and non-inflammatory lesion counts and overall improvement scores. The study cohort was randomized into two groups: the nadifloxacin group and the clindamycin group. All participants were instructed to apply a thin layer of medication on acne lesions; 2.5% benzoyl peroxide gel was used once daily before bedtime, and either 1% clindamycin gel or 1% nadifloxacin gel was applied twice daily. The efficacy was analyzed by comparing changes in total, inflammatory, and non-inflammatory lesion counts, and patients rated overall improvement using a six-point scale (worsened, no improvement, slight improvement, moderate improvement, good improvement, cleared) at the end of the treatment. The number of lesions in both groups gradually decreased. Lesion count analysis showed no significant difference between the two groups. However, more patients in the nadifloxacin and benzoyl peroxide group reported good improvement compared to the clindamycin and benzoyl peroxide group. No lesion deterioration was reported in either group, and no patient reported complete clearance. Two adverse events, burning sensation and dryness, were reported during the study, with no significant difference in adverse event incidence between the groups. Topical nadifloxacin is a novel fluoroquinolone drug that is effective, well-tolerated, and safe for treating mild to moderate facial acne. Its clinical efficacy is comparable to clindamycin, making it a viable adjunctive treatment with benzoyl peroxide.
Acne vulgaris is a chronic inflammatory disease of the pilosebaceous unit, a common skin condition affecting at least 85% of adolescents and young adults. Topical retinoids (e.g., tretinoin) are commonly used in combination with antibiotics (e.g., clindamycin (CLN) and nadifloxacin (NAD)) to treat mild to moderate acne vulgaris. So, can you use nadifloxacin and clindamycin together?
Nadifloxacin is typically used topically, while clindamycin can be used both topically and systemically. Since nadifloxacin is applied directly to the skin, systemic interactions are less of a concern. There are no known direct interactions between topical nadifloxacin and systemic or topical clindamycin. However, the combination should be monitored to ensure effective treatment of the infection and minimize potential adverse reactions.
Common side effects include nausea, vomiting, diarrhea, rash, and pain at the injection site. It increases the risk of hospital-acquired Clostridium difficile colitis by about fourfold, so it is recommended only when other antibiotics are unsuitable.
During treatment, some patients may experience side effects, mainly affecting the skin and subcutaneous tissues. Common reactions include burning sensations and itching, which are the most common side effects. Patients may also experience contact dermatitis, skin dryness, and skin irritation.
In specific cases of combined use, unnecessary side effects or resistance may occur.
In certain cases, combining antibiotics may be beneficial for broad-spectrum treatment or targeting different bacteria. Nadifloxacin and clindamycin are both antibiotics used to treat bacterial infections, but their mechanisms of action and activity spectra differ. Whether they can be used together depends on the specific clinical situation, type of infection, and healthcare provider's recommendation.
One of the main uses of topical antibacterial drugs is the treatment of acne, with benzoyl peroxide being the drug of choice, used alone or in combination with antibiotics or retinoids. Mupirocin and fusidic acid are two of the most commonly used antibiotics for treating superficial impetigo and eradicating staphylococcal carriage.
Although nadifloxacin has been proven effective in treating skin and soft tissue infections (SSTI), there is limited data comparing its efficacy and safety with other antimicrobials. Janbandhu et al. compared the safety and efficacy of nadifloxacin and mupirocin in children with SSTI. The study was a single-center, open-label, randomized, parallel-group comparison involving 60 children under 12 years old with SSTI. The trial group (n=30) received 1% nadifloxacin ointment, and the reference group (n=30) received 1% mupirocin ointment, applied twice daily. Patients were followed up on days 4, 8, and 15. The efficacy of the study drugs was evaluated by clinical and bacteriological cure rates. Safety was assessed by reporting adverse events.
The study concluded that nadifloxacin and mupirocin were equally effective and safe for treating SSTI in the Indian pediatric population.
Although nadifloxacin and clindamycin are both effective antibiotics, they have significant differences in mechanisms of action, indications, and side effects. Therefore, when choosing the right antibiotic treatment, patients should thoroughly understand their specific situation and medical needs. It is strongly recommended that you consult a doctor before using these drugs to obtain professional advice and personalized treatment plans, ensuring safety and optimal efficacy.
[1]https://en.wikipedia.org/wiki/Clindamycin
[2]https://en.wikipedia.org/wiki/Nadifloxacin
[3]https://discovery.researcher.life/article/safety-and-efficacy-of-nadifloxacin-versus-clindamycin-observed-in-a-tertiary-care-teaching-hospital-of-deccan-plateau/2224bfac83e33b7ab6eb459feca84480
[4]https://pesquisa.bvsalud.org/portal/resource/pt/sea-204446
[5]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8208253/
[6]Janbandhu S, Chaudhary S, Chaudhary S, et al. An evaluation of safety and efficacy of nadifloxacin 1% ointment versus mupirocin 1% ointment in Indian children with skin and soft tissue infection[J]. 2020.
[7]Gang Lixia, Wang Sheng. Study on analytical methodology of nadifloxacin related substances[J]. Mechanical and Electrical Information, 2014, (29): 37-42. DOI: 10.19514/j.cnki.cn32-1628/tm.2014.29.008.
 |
 |