
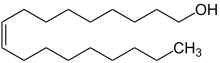
Oleyl alcohol, also known as octadec-9-en-1-ol or cis-9-octadecen-1-ol, is a non-ionic unsaturated fatty alcohol with a double bond at the 9th position (Z-isomer). As a non-ionic surfactant and metabolite, oleyl alcohol belongs to the long-chain primary fatty alcohol class, with a fatty alcohol ratio of 18:1.
It is a naturally occurring long-chain aliphatic alcohol found in fish oil and is extensively used in textile softening and lubrication. Additionally, oleyl alcohol is applied in the production of carbon paper, wax paper, and printing inks. It is also used as an antifoaming agent and cutting lubricant, and serves as a precursor for the preparation of sulfate esters. These esters are widely used in detergents and wetting agents. Oleyl alcohol is present in various personal care products, including conditioners, foundations, eye cosmetics, moisturizers, and cleansers.
Oleyl alcohol plays a role in multiple areas, including the manufacture of detergents, wetting agents, antifoaming agents, metal cutting lubricants, plasticizers, and fabric softeners. It is also used as a raw material in organic synthesis and specialty surfactants, contributing to the production of oil additives, cold-resistant plasticizers, lubricants, and solvents.
Oleyl alcohol and its derivatives exhibit good biodegradability and surface activity, making them suitable for use in the detergent and textile industries. Compared to saturated alcohols, oleyl alcohol, oleyl alcohol ethers, and oleyl alcohol sulfates have lower Krafft temperatures and excellent wetting properties, as well as low surface tension. Oleyl alcohol sulfate demonstrates effective cleaning, washing, and foaming properties with minimal skin irritation, making it especially suitable for cold water washing. Additionally, oleyl alcohol sulfate is a high-performance new surfactant that can replace AE and AES surfactants and represents one of the ways to develop and utilize high-carbon fatty alcohol resources.
Oleyl alcohol is also investigated as a carrier for drug delivery through the skin or mucous membranes. It is commonly used as a penetration enhancer and permeation promoter to facilitate the delivery of topical medications.
In cosmetics, oleyl alcohol functions as a non-ionic surfactant and is widely used in shampoos and conditioners as a hair coating agent. In creams and lotions, it acts as an emollient (improving skin softness), emulsifier, and thickener.
In 1960, three cases of lip dermatitis caused by oleyl alcohol were described. All products reacted with high-purity oleyl alcohol (100%) present in the cosmetic base. Patients also reacted to oleyl alcohol at 5% concentration in liquid paraffin. Another patient was considered to react to impurities in commercial stearyl alcohol, with oleyl alcohol being one of the most likely allergens.
Stearyl alcohol, oleyl alcohol, and octyl dodecanol are long-chain saturated or unsaturated (oleyl-based) fatty alcohols. They are used in concentrations ranging from less than 0.1% to more than 50% in various cosmetic categories. Studies have described the metabolism of stearyl alcohol and oleyl alcohol in rats. Acute oral toxicity studies indicate very low toxicity levels. In rabbit irritation tests, these alcohols cause minimal eye irritation and minimal to mild skin irritation. Clinical patch tests show very low potential for skin irritation and sensitization. Photoreactivity studies of products containing these ingredients show negative results for phototoxicity or photosensitivity.
A. Tosti et al. investigated the frequency of sensitization to fatty alcohols in a group of suspected cases of cosmetic or drug contact dermatitis. From May 1992 to September 1995, a series of patch tests with five Italian alcohols were conducted on 140 patients. Among the 25 women and 9 men, aged between 14 and 72 years, positive patch tests for fatty alcohols were observed. Thirty-three of these were for oleyl alcohol. A total of 39 reactions were detected, with 5 patients showing more than one positive reaction. The findings indicate that sensitization to oleyl alcohol is not uncommon among patients with contact dermatitis caused by cosmetics or topical medications.
At high concentrations, oleyl alcohol may cause minor skin irritation in some individuals.
Oleyl alcohol may cause irritation upon contact with the eyes.
Oleyl alcohol has a moderate flash point, indicating flammability in high-temperature environments.
Consult a physician. Provide the safety data sheet to the attending doctor.
If inhaled, move the patient to fresh air. Perform artificial respiration if breathing stops. Consult a physician.
Wash with soap and plenty of water. Consult a physician.
Rinse thoroughly with water for at least 15 minutes and consult a physician.
Do not give anything by mouth to an unconscious person. Rinse the mouth with water. Consult a physician.
Basic treatment: Establish an open airway (if necessary, establish an oropharyngeal or nasopharyngeal airway). Perform suction if needed. Monitor for signs of respiratory distress and assist ventilation if necessary. Administer oxygen through a non-rebreather mask at a flow rate of 10 to 15 L/min. Monitor for pulmonary edema and treat if necessary. Monitor for shock and treat if necessary. Anticipate seizures and treat if necessary. If the eyes are contaminated, immediately rinse with water. Continue rinsing each eye with 0.9% saline during transport. Do not use emetics. If the patient has difficulty swallowing or a strong gag reflex, rinse the mouth and dilute with water (5 ml/kg to 200 ml). After cleansing, cover the burn area with dry, sterile dressings.
Avoid contact with skin and eyes. Avoid creating dust and aerosols. Avoid contact—obtain special instructions before use. Provide adequate ventilation in areas where dust is formed.
Store in a cool place. Keep the container tightly closed and store in a dry, well-ventilated area.
[1]Elder, R L. Final report on the safety assessment of stearyl alcohol, oleyl alcohol, and octyl dodecanol[J]. J Am Coll Toxicol, 1985, 4(5): 1-29.
[2]Tosti A, Vincenzi C, Guerra L, et al. Contact dermatitis from fatty alcohols[J]. Contact Dermatitis, 1996, 35(5): 287-289.
[3]https://pubmed.ncbi.nlm.nih.gov/37950377/
[4]https://en.wikipedia.org/wiki/Oleyl_alcohol
[5]https://baike.baidu.com/item/%E6%B2%B9%E9%86%87
[6] Chang Ying, Wang Chenghe, Liu Yanjun. Synthesis and application of oleyl alcohol[C]. //2003 Surfactant Technology and Economics Collection. Dalian: Fine and Specialty Chemicals Editorial Department, 2003:199-203.
[7] Liu Yanjun, Chang Ying. Synthesis and application of oleyl alcohol[J]. Fine and Specialty Chemicals, 2004, 12(5): 8-11. DOI: 10.3969/j.issn.1008-1100.2004.05.002.
[8]Tada, J., Toi, Y., & Arata, J. (1994). Atopic dermatitis with severe facial lesions exacerbated by contact dermatitis from topical medicaments. Contact Dermatitis, 31. https://doi.org/10.1111/J.1600-0536.1994.TB02002. X.
[9]https://drugs.ncats.io/substance/172F2WN8DV
[10]https://pubchem.ncbi.nlm.nih.gov/compound/Oleyl_alcohol
 |
 |
 |