
Pemetrexed disodium hemipentahydrate, chemically known as N-[4-[2-(2-amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]-L-Glutamic acid disodium salt, 2.5 Hydrate (N-[4-[2-(2-amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]-L-Glutamic acid disodium salt, 2.5 Hydrate), has the following structure:
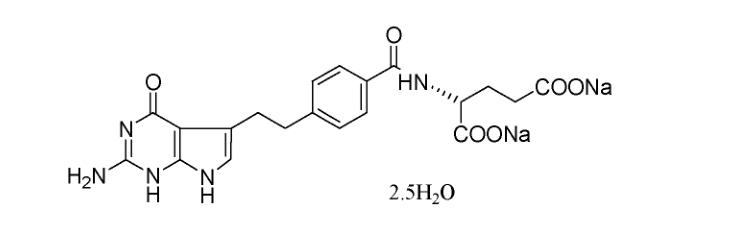
Pemetrexed disodium hemipentahydrate is a novel folate metabolism inhibitor with a pyrrolo[2,3-d]pyrimidine structure that simultaneously inhibits thymidylate synthase (TS), dihydrofolate reductase (DHFR), and GARFT, making it a multi-target folate antagonist. By interfering with folate-dependent metabolic pathways, it fundamentally inhibits cell replication, exerting its antitumor activity. Pemetrexed disodium hemipentahydrate is transported into cells via the reduced folate carrier (RFC) and folate transport membrane proteins (FPB). Once inside the cell, pemetrexed disodium hemipentahydrate is rapidly catalyzed by folate polyglutamate synthetase (FPGS) to form polyglutamate compounds. Both non-polyglutamated and polyglutamated pemetrexed disodium hemipentahydrate act as competitive inhibitors, affecting the activity of several folate-dependent enzymes, including key enzymes in nucleotide biosynthesis. This mechanism is similar to the inhibitory effects of methotrexate on TS and DHFR. Pemetrexed disodium hemipentahydrate and its polyglutamate derivatives competitively bind to various folate synthesis-related enzymes, which play critical roles in thymidylate and purine nucleotide biosynthesis. Due to its inhibition of multiple targets, pemetrexed disodium hemipentahydrate enhances its antitumor activity and has a lower likelihood of resistance development.
Pemetrexed disodium hemipentahydrate, as a multi-target folate antagonist, has several advantages: (1) the drug targets multiple sites, reducing the likelihood of resistance; (2) the inhibition of multiple targets in the folate metabolism pathway during DNA synthesis may have additive or even synergistic effects; (3) differences in folate metabolism enzyme activities among various tumor cells affect the efficacy of anti-folate drugs, with multi-target drugs being less affected by these differences compared to single-target drugs. Based on the unique mechanism of action of pemetrexed disodium hemipentahydrate and its clinical research results expanding its indications, the drug has significant potential in the field of antitumor therapy and a broad prospect in tumor treatment applications.
The common side effects of pemetrexed disodium hemipentahydrate include hematologic toxicity, primarily neutropenia, which is often dose-limiting. Other frequent adverse effects include low white blood cell count, nausea, vomiting, fatigue, rash, and diarrhea. Additional adverse effects may include liver and kidney function abnormalities, vomiting, constipation, and chest pain. These symptoms may be indicative of bone marrow suppression-related infections caused by the drug.
While pemetrexed disodium hemipentahydrate demonstrates significant efficacy in cancer treatment, its use must be tailored to individual patient conditions. Patients should always consult their physician when using this drug to ensure that the treatment plan is safe and effective, and to adjust the dosage and therapy according to medical advice. Professional medical guidance helps maximize the drug's therapeutic effects while minimizing potential side effects.
[3]https://go.drugbank.com/salts/DBSALT001782
[4]https://baike.baidu.com/item/%E5%9F%B9%E7%BE%8E%E6%9B%B2%E5%A1%9E%E4%BA%8C%E9%92%A0
 |