
Peri acid, chemically known as 8-amino-1-naphthalenesulfonic acid or 1-naphthylamine-8-sulfonic acid, has the molecular formula C10H9NO3S and a molecular weight of 223.25. It appears as white needle-like crystals with a melting point >350°C, is slightly soluble in water, and readily dissolves in glacial acetic acid. Peri acid is a crucial intermediate in the dye industry, used in the production of a range of naphthalene dyes. It is also utilized to prepare other dye intermediates such as phenyl peri acid and toluene peri acid, as well as various active and reductive dyes. Its structural formula is:

Peri Acid structural
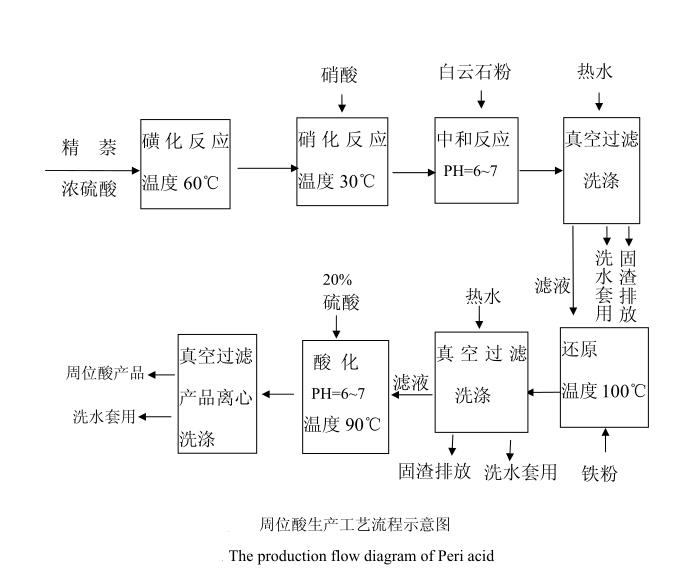
The production process involves mixing concentrated sulfuric acid with purified naphthalene, followed by heating in a sulfonation kettle for the sulfonation reaction. After the reaction, nitric acid is used for nitration, and then the product is neutralized with a limestone solution. The neutralized product is vacuum-filtered, and the filtrate is reduced with iron powder. The reduced product is filtered again, and the filtrate is acidified with 20% sulfuric acid to precipitate peri acid. Finally, centrifugation is used to obtain the peri acid product. The specific process flow is illustrated in the diagram below:
Sulfonic acid functional groups are strong acid catalysts widely used in various chemical reactions. However, in many reactions where water is a byproduct, the catalytic activity and selectivity may be unsatisfactory due to the hydrophilic SO3H sites being affected by decreased acidity and undergoing hydrolytic side reactions through water adsorption. A novel hydrophobic aromatic sulfonic acid functionalized biochar has been successfully prepared through a one-pot diazotization reduction method using biochar with amino aromatic sulfonic acids (such as 4-amino-benzenesulfonic acid, 4-amino-1-naphthalenesulfonic acid, 8-amino-1-naphthalenesulfonic acid, and 4-amino-3-hydroxy-1-naphthalenesulfonic acid). This biochar exhibits a large specific surface area of 200-400 m2/g, a hydrophobic network with a water contact angle above 120°, and a sulfonic acid concentration of over 1.0 mmol/g. The catalyst is stable and shows excellent cyclic performance after 6 runs. The successful preparation of hydrophobic biochar-based acidic catalysts not only provides a new avenue for the high-value utilization of biochar but also eliminates the negative impact of water on many catalytic reactions.
In industrial production, peri acid is obtained from purified naphthalene through sulfonation, nitration, reduction, and acid precipitation. During its production, highly acidic organic wastewater is generated, which can reach thousands of times in color and a chemical oxygen demand (COD) value of tens of thousands of milligrams per liter. Direct discharge can severely pollute water bodies and cause environmental harm. Huang Jun and others used microfiltration technology to pre-treat peri acid organic wastewater, followed by secondary treatment with macroporous adsorption resins. They studied the effects of resin types, adsorption flow rates, adsorption temperatures, and desorption agents on the adsorption and desorption performance of the resin. Suitable adsorption resins (H-103) and optimal adsorption and desorption conditions were identified. The presence of peri acid in wastewater has led to the development of these treatment methods.
Peri acid is used to manufacture a series of naphthalene dyes, such as C.I. Acid Black 35, C.I. Reactive Orange 35, and Sulfur Dark Green 3GW. It is also utilized in preparing other dye intermediates like phenyl peri acid, toluene peri acid, amino C acid, and Chicago acid. Additionally, it is used in the production of various active and reductive dyes.
The molecular structure and properties of peri acid enhance efficiency and effectiveness. As a catalyst or reagent, peri acid can accelerate reaction rates, increase product yields, and improve selectivity. Compared to traditional alternatives, peri acid often demonstrates superior performance in reaction time, energy consumption, and by-product formation. These advantages can lead to more sustainable and cost-effective chemical processes.
Peri acid, with its unique chemical properties and wide application potential, offers significant product advantages. For more information on peri acid supply and product details, it is recommended that readers visit the Guidechem website, where they can find reliable suppliers and make purchases. Through Guidechem, you can easily obtain peri acid and further explore its potential in various applications.
 |