
Phenazine derivatives hold a significant place in medicinal chemistry due to their wide range of biological activities. The notable pharmacological properties of natural phenazines have driven the design and development of methods to obtain synthetic analogues with similar or even better modified properties. Various synthetic phenazine analogues are renowned for their antimalarial, antitrypanosomal, anti-hepatitis C virus replication, antitumor, antifungal, antileishmanial, and insecticidal activities. Phenazines are also used as electroactive materials in organic field-effect transistors (OFETs), organic light-emitting diodes (OLEDs), dye-sensitized solar cells, and photocatalytic photoactive materials, as well as fluorescent tracers and dyes for subcellular components and biological events. Additionally, fluorescent phenazines can serve as photosensitizers in photodynamic therapy (PDT), where the combination of light and photosensitizers generates highly reactive oxygen or hydroxyl radicals near tumors, selectively destroying target tissues.
Due to the extensive applications of the phenazine scaffold in medicine and industry, it has become an attractive synthetic target. Common methods for phenazine synthesis include Wohl-Aue method, Beirut method, condensation of 1,2-diaminobenzene with 2C units, reduction cyclization of diphenylamine, oxidation cyclization of 1,2-diaminobenzene/diphenylamine, Pd-catalyzed N-arylation, and multicomponent methods.
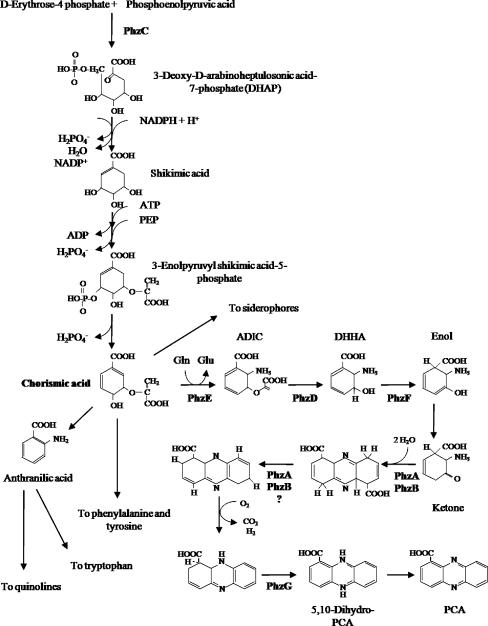
Here is a brief overview of phenazine biosynthesis. Phenazine originates from the shikimic acid pathway, using intermediate branch acids as fundamental biosynthetic branch points for phenazine aromatic structures. The shikimic acid biosynthetic pathway is highly conserved and involved in the production of various metabolites required for primary growth, including three aromatic amino acids and p-aminobenzoic acid. In bacteria, this pathway is tightly regulated, primarily occurring at the first step where 3-deoxy-D-arabino-heptulosonate-7-phosphate (DAHP) synthase catalyzes the condensation of erythrose-4-phosphate and phosphoenolpyruvate. Many bacteria possess type I DAHP synthase isoenzymes, each subject to feedback inhibition by one of the pathway's final products. Interestingly, in various phenazine-producing microorganisms, the type II-3-DAHP synthase encoded by the phzC biosynthetic operon exhibits enzyme characteristics closer to that of DAHP synthase in Solanaceae plants rather than prokaryotic DAHP synthases. In plants, type II-3-DAHP synthase lacks the regulatory cyclic structure and is not regulated by the three aromatic amino acid products. However, its activity increases during specific plant growth stages, such as seed germination, branch growth, and glyphosate exposure. The expression of phzC may help enhance the first condensation step, ensuring a sufficient supply of substrates required for phenazine synthesis. However, the unregulated expression of phzC might deplete essential metabolites needed for primary growth, potentially lowering the overall adaptability of the rhizosphere. This demand for controlled expression at least partially explains the complex regulation of phenazine production.

Traditionally, the synthesis of phenazines and their derivatives used chemical methods involving toxic organic solvents, hazardous reagents, and the risk of harmful metal contamination in the final products. These drawbacks have severely limited the widespread application of phenazine derivatives in therapeutic and pharmaceutical industries. Consequently, there is an increasing need for environmentally friendly methods to address these challenges and reduce environmental damage. Nowadays, green and efficient methods can be employed to synthesize phenazines and their derivatives. These methods include mechanical synthesis, solvent-free and catalyst-free synthesis, green solvent synthesis, ultrasound-assisted synthesis, microwave-assisted synthesis, and other similar approaches, given the widespread presence of phenazine backbones as bioactive components and the growing demand to reduce hazardous solvents, catalysts, and energy use.
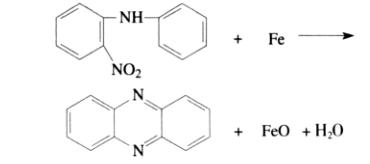
Henry reacted N-phenyl-2-nitroaniline in the presence of iron powder to obtain phenazine with a yield of 46%. The nitro group is first reduced to an amino group, followed by an oxidative cyclization reaction to produce phenazine.
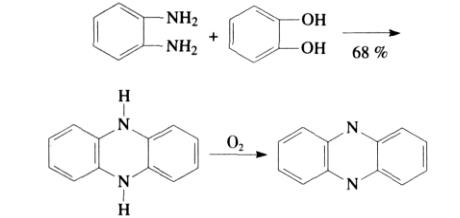
Morley et al., based on earlier research methods, synthesized phenazine and its derivative 5,10-dihydrophenazine by reacting catechol with ortho-diaminobenzene. The process was conducted in a sealed container at 200 to 210°C for 35 to 40 hours under applied pressure. The intermediate product was 5,10-dihydrophenazine, with a crude yield of approximately 60% after purification. This intermediate was then converted to phenazine by passing oxygen through it. However, the main drawback of this method is its difficulty in large-scale production and the generation of numerous undetectable by-products.
Wataya used 2-nitroaniline or its substituted compounds, undergoing cyclization in dimethyl sulfoxide with strong alkaline metal alcoholates. Dimethyl sulfoxide is a relatively expensive solvent, whereas other solvents like benzene, toluene, ethers, and hydroxy compounds such as aliphatic alcohols either fail, react slowly, or yield low results. The products obtained using dimethyl sulfoxide are difficult to purify and require column chromatography, which increases the production costs of chemical synthesis.
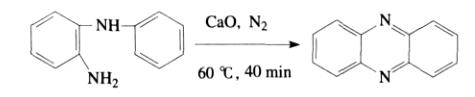
Horaguchi et al. discovered that 2-amino-diphenylamine could be reacted at 600°C for 40 minutes in the presence of calcium oxide as a catalyst under nitrogen protection to achieve a 70% yield of phenazine.
 |
 |
 |