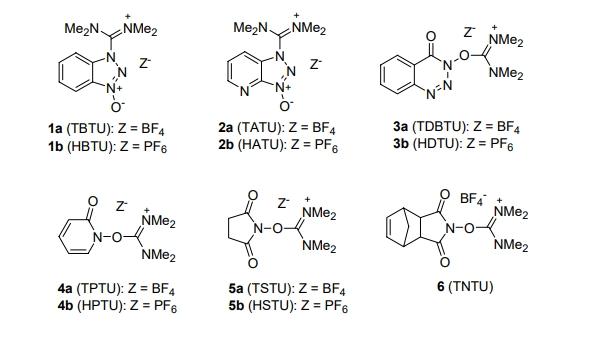
N, N, N, N-Tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate (TSTU) is an effective coupling reagent. TSTU is frequently used in coupling reactions of biomolecules such as proteins, peptides, and oligosaccharides. It forms active ester intermediates with carboxylic acids and rapidly reacts with amines in aqueous solutions to construct stable amide bonds. TSTU has widespread applications in glycopeptide synthesis, oligosaccharide-protein conjugation, and fluorescent labeling. Notably, the combination of TSTU with N-hydroxysuccinimide and CuCl2 effectively suppresses racemization of carboxyl-terminal N-methyl amino acids in peptide synthesis. The structure of TSTU is shown below:

TSTU's usage in peptide synthesis is extensive, serving as a coupling reagent. TSTU activates carboxylic acids, making them more susceptible to nucleophilic attack by amines, thereby promoting peptide bond formation. TSTU is an efficient coupling agent that swiftly activates carboxylic acids to form N-succinimidyl active esters, which then react efficiently with primary amines to produce amide bonds. Even in aqueous environments or the presence of alcohols, TSTU maintains high reactivity. Additionally, TSTU effectively inhibits racemization when coupling protected α-amino acids.
Xinhua He et al. reported an efficient method for synthesizing (Z)-3-ylidenephthalides at room temperature via TSTU-mediated intramolecular cyclization of readily accessible 2-acylbenzoic acids. This method yields various substituted (Z)-3-ylidenephthalides in good to excellent yields, with notable application in gram-scale preparation of antiplatelet drug n-butyl phthalide.
TSTU is a superior reagent for forming carboxamide bonds, effectively converting carboxylate-derived oligosaccharide derivatives into active esters and glyconjugates. Mats Andersson et al. synthesized glucosides and lactose glycosides with diethylene glycol and tetraethylene glycol spacers, converting them into bifunctional (alcohol, ester) spacer molecules. After deprotection, these free carboxyl-containing spacer glycosides were efficiently converted into glyconjugates using N, N, N, N-tetramethyl(O-succinimidyl)uronium tetrafluoroborate (TSTU), forming active esters.
New polymeric urea salts P-TSTU and P-HSTU, synthesized from polymeric N-hydroxysuccinimide (P-HOSu), serve as solid-supported reagents for peptide coupling reactions. The released P-HOSu can be recovered through simple filtration and reused for preparing new reagents.
Mayke Werner et al. demonstrated the successful synthesis and solid-state NMR characterization of silica-based organic-inorganic hybrid materials. Collagen-like peptides were immobilized on carboxyl-functionalized mesoporous silica (COOH/SiOx) materials. TSTU was used to preactivate the silica material, allowing the peptide to covalently bind with the linker. The decrease in 13C CP-MAS NMR signals of the TSTU portion indicated successful covalent attachment. 15N CP-MAS dynamic nuclear polarization (DNP) qualitatively distinguished covalently bound peptides from adsorbed peptides, with the 15N signal of the peptide N-terminus showing a clear downfield shift indicating the binding site. DNP enhancement techniques detected naturally abundant 15N nuclei without expensive peptide labeling.
A novel coupling system composed of TSTU, HOSu, and CuCl completely eliminates racemization of N-methyl amino acid residues during segment coupling.
TSTU is a chemical substance with potential health risks. It is labeled with the GHS07 warning icon, indicating that it may cause skin and eye irritation. To minimize exposure and prevent accidents, handle TSTU with care and take appropriate safety measures. Always wear protective gloves, goggles, and a lab coat when handling this compound. Work with TSTU in a well-ventilated area and avoid inhaling its vapors. Store it in a safe, labeled container, away from incompatible substances. If accidental contact or ingestion occurs, seek medical attention immediately.
 |
 |