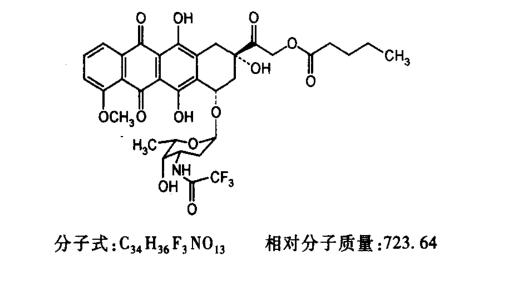
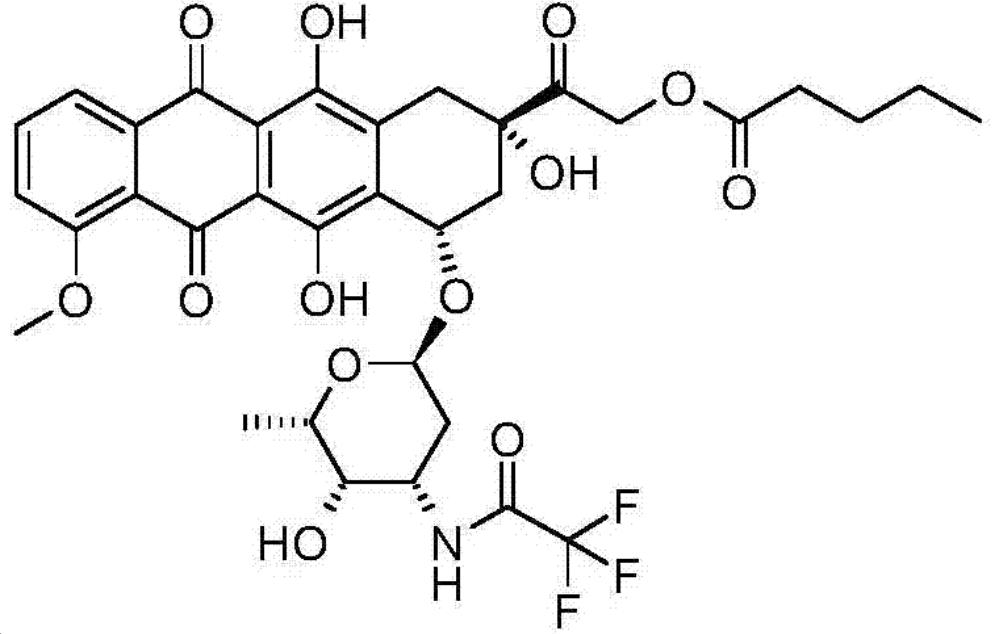
Generic name: Valrubicin; Brand names: Valstar, Valtaxin; Chemical name: (2S-cis)-2-[1,2,3,4,6,11-hexahydro-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-4-[[2,3,6-trideoxy-3-[(trifluoroacetyl)amino]-α-L-lyxo-hexopyranosyl]oxy]-2-naphthacenyl]-2-oxoethyl pentanoate. This compound is an orange-yellow or orange-red powder with a melting point of 135-136°C. It has strong lipophilicity, dissolving in dichloromethane, ethanol, methanol, and acetone, but is almost insoluble in water. As an anthracycline drug, its primary action is to interfere with the normal DNA division and recombination, affecting various biological functions of cells, mainly by disrupting nucleic acid metabolism. It easily penetrates cells and inhibits the formation of nucleotides, thereby causing significant chromosomal damage and halting the cell cycle at the G2 phase.
Valrubicin is used to treat a type of bladder cancer (carcinoma in situ; CIS) that cannot be effectively treated with other medications (BCG therapy) and is suitable for patients who cannot immediately undergo a full or partial cystectomy, where bladder removal surgery would lead to unacceptable morbidity or mortality. However, only about one in five patients respond to Valrubicin treatment, and delaying bladder surgery could result in the spread of bladder cancer, which may be life-threatening. Valrubicin is an anthracycline antibiotic used only in cancer chemotherapy. It can slow or stop the growth of cancer cells in the body.
Valrubicin is an anthracycline drug. It has been noted that anthracycline antibiotics play a subtle role in the metabolism and disposition of these non-specific cell cycle inhibitors used in cancer chemotherapy. These compounds are derived from streptomyces, including doxorubicin, daunorubicin, epirubicin, idarubicin, Valrubicin, and mitoxantrone.
In the 1960s, Di Marco and colleagues at the Farmitalia Research Laboratories first described daunorubicin. They isolated daunorubicin from the bacterium S. peucetius. Daunorubicin was later renamed daunorubicin (Dau), a combination of daunorubicin and doxorubicin, the latter being the same compound isolated and identified by Dubost and colleagues from Rhone-Poulenc from S. coerubleorubidus. Dau’s initial clinical trials on solid tumors were not very successful, but its application in lymphomas achieved remarkable results. This prompted research into other similar molecules.
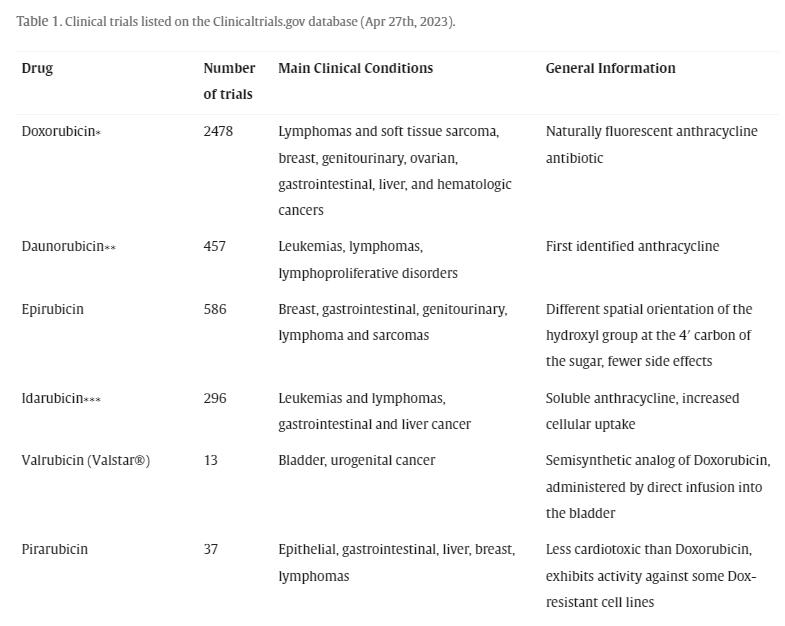
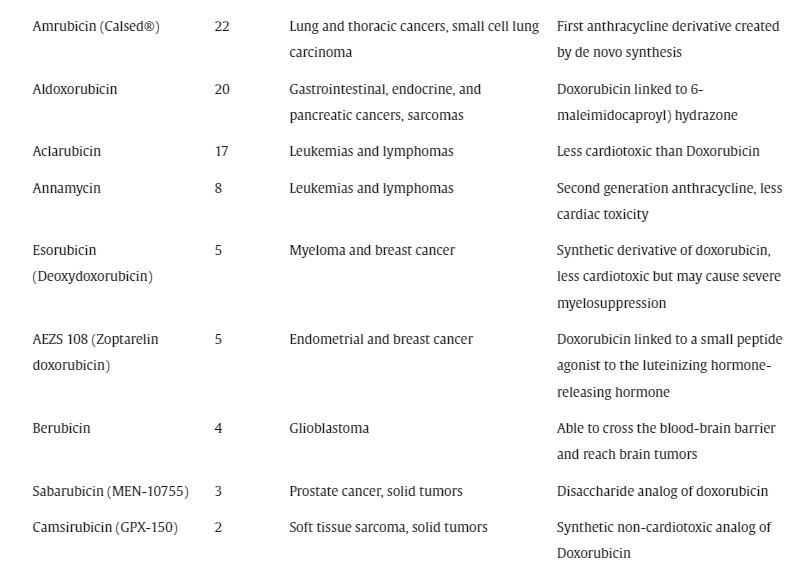
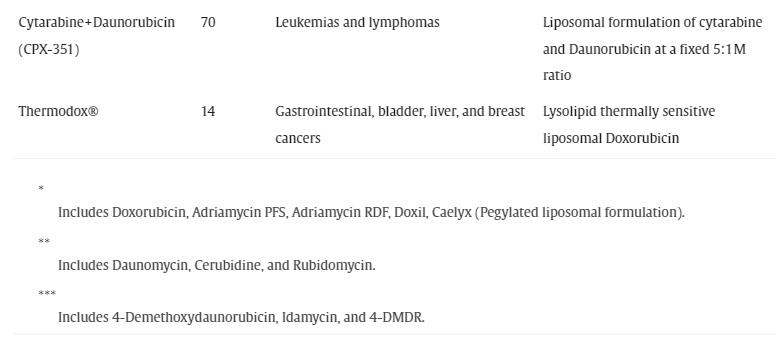
Shortly thereafter, Farmitalia discovered doxorubicin. It was produced using a mutated strain of the bacterium S. peucetius var. caesius. This molecule (also sold under the name doxorubicin) showed impressive activity against a wide range of solid tumors in animals and clinical trials and was quickly implemented in clinical settings. Despite the discovery of anthracycline drugs over 50 years ago, they continue to be widely used in various solid tumors and hematological cancers. In fact, these drugs remain among the most effective anticancer agents to date. Their importance can be seen in the large number of clinical trials registered on the clinicaltrial.gov website as of April 27, 2023, as shown in the table below:
Valrubicin was approved in 1998 and was initially marketed under the brand name Valstar in the late 1990s for intravesical therapy in BCG-refractory cancer patients at risk of morbidity or mortality from cystectomy (Steinberg et al., 2000). Due to manufacturing issues, the drug was withdrawn from the market in 2002 and reintroduced in 2009. While most anthracycline drugs exhibit significant anticancer activity as single agents, they are often used in combination chemotherapy or nanodrug formulations to enhance therapeutic efficacy and reduce toxicity.
Michael S. Cookson et al. conducted a U.S. multicenter, retrospective medical record study examining the effectiveness, safety, and patterns of clinicians using Valrubicin for treating non-muscle invasive bladder cancer (NMIBC) since its reintroduction in 2009. The medical records of 113 patients met the inclusion criteria; 100 patients (88.5%) completed Valrubicin treatment. The median age was 75 years (range 42-95 years). The median duration of NMIBC since diagnosis was 31 months: 51.3% (58/113) of patients had CIS only, 31.9% (36/113) had unspecified NMIBC. Most patients (94.7% (107/113)) received more than three instillations of Valrubicin, and 70.8% (80/113) completed the entire course. The EFS rates (95% confidence interval) at 3, 6, and 12 months were 51.6% (40.9-61.3%), 30.4% (20.4-41.1%), and 16.4% (7.9-27.5%), respectively. The median time to the event was 3.5 (2.5–4.0) months after the first instillation of Valrubicin. 49.6% (56/113) of patients experienced local adverse reactions (LAR); most LARs were mild (93.6%). The most common LARs were hematuria, urinary frequency, urgency, bladder spasms, and dysuria. A total of 4.4% (5/113) of patients discontinued Valrubicin due to adverse events or LARs.
These trials demonstrate the efficacy and tolerability of Valrubicin in certain CIS patients before considering cystectomy.
Valrubicin plays a crucial role in the treatment of bladder cancer and shows promising potential in antitumor therapy. However, every patient’s condition and needs are unique, so it is essential to consult your doctor before using this drug to obtain professional advice and guidance to ensure the most suitable treatment plan is selected.
[1] https://www.sciencedirect.com/science/article/pii/S0098299723000456
[2] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4144261/
[3] sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/valrubicin
[4] Zhang Li, Qiu Guofu. Valrubicin [J]. Chinese Journal of New Drugs, 2006, 15(13): 1123-1124. DOI: 10.3321/j.issn:1003-3734.2006.13.030.
[5] https://go.drugbank.com/drugs/DB00385
[6] https://medlineplus.gov/
[7] https://www.wikidoc.org/index.php/Valrubicin
 |
 |
 |