Panaxadiol is a dammarane-type triterpene saponin with pharmacological activities including cytotoxicity, anti-dementia, anti-inflammatory, and cardiovascular protection. Ginseng (Panax ginseng) is a popular plant, belonging to the Araliaceae family, with about 11 species in the Panax genus. The term "panax" originates from Greek, meaning "all-healing" or "panacea."

Ginsenosides are a group of saponins and the main functional components of ginseng. They are categorized into two groups: tetracyclic dammarane and pentacyclic oleanane. Dammarane can be further divided into two types: protopanaxadiol (PPD) (e.g., Rb1, Rb2, Rc, Rd, Rg3, Rh2, Rh3) and protopanaxatriol (PPT) (e.g., Rg1, Re, Rf, Rg2, Rh1). Panaxadiol, a pseudo-sapogenin of dammarane-type saponins (as shown in the image below), represents an emerging class of anti-tumor agents with various biological properties such as anti-cancer, anti-emetic, and radioprotective effects.
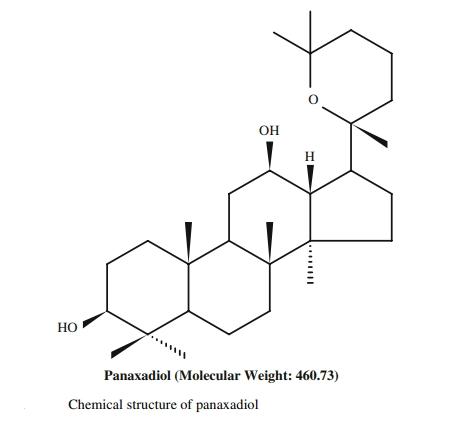
Panaxadiol has been evaluated for cytotoxicity and showed minimal cell toxicity while significantly inhibiting the expression of PD-L1 at the protein and mRNA levels in a dose-dependent manner. Additionally, Panaxadiol inhibits hypoxia-induced hypoxia-inducible factor (HIF)-1α synthesis through the phosphoinositide 3-kinase (PI3K) and mitogen-activated protein kinase (MAPK) pathways, without affecting HIF-1α degradation. Panaxadiol also suppresses STAT3 activation through JAK1, JAK2, and Src pathways. Furthermore, pre-treatment with Panaxadiol enhances the activity of cytotoxic T lymphocytes (CTLs) and restores their ability to kill tumor cells. Immunoprecipitation studies suggest that Panaxadiol inhibits PD-L1 expression by blocking the interaction between HIF-1α and STAT3. Colony formation and EdU labeling assays further demonstrate Panaxadiol’s inhibitory effect on tumor proliferation. In vivo xenograft studies also confirm the anti-proliferative effects of Panaxadiol.
The retinoid-related orphan receptor γ (RORγ) is a key transcription factor for pro-inflammatory IL-17A production. Research by Si-Yu Tian and colleagues revealed that RORγ deficiency protects mice from STZ-induced type 1 diabetes (T1D) by inhibiting IL-17A production, thereby improving pancreatic β-cell function and highlighting a potential new therapeutic target for T1D. The study identified a novel RORγ inverse agonist—Panaxadiol derived from ginseng—which selectively inhibits RORγ transcriptional activity and differs in cofactor recruitment from known RORγ ligands. Structural and functional studies of receptor-ligand interactions revealed the molecular basis for Panaxadiol’s unique binding mode in the RORγ ligand-binding pocket. Despite its inverse agonist activity, Panaxadiol induces a typical active conformation in the C-terminal AF-2 helix of RORγ. Interestingly, Panaxadiol improves STZ-induced type 1 diabetes in mice by inhibiting IL-17A production in an RORγ-dependent manner.
Panaxadiol exhibits anti-aging and inhibitory mechanisms. Experiments using indicators such as lipid peroxidation, lipofuscin, superoxide dismutase, and catalase showed that Panaxadiol significantly inhibits lipid peroxide formation in the human brain and liver, effectively reduces lipofuscin levels in the liver, and increases superoxide dismutase and catalase levels in plasma.
In a rat model of hyperglycemia induced by low-dose streptozotocin injection, adrenaline, and high-fat diet, Panaxadiol saponins were administered. Results showed that Panaxadiol at doses of 50, 100, and 200 mg/kg significantly reduced hyperglycemia induced by subcutaneous adrenaline and intravenous streptozotocin, and notably increased liver glycogen content.
Kyeong T and colleagues studied the anti-nausea and vomiting effects of Panaxadiol. Nausea and vomiting are commonly reported side effects in patients undergoing general anesthesia and cancer patients receiving chemotherapy or radiotherapy. The 5-HT3α receptor is known to mediate nausea and vomiting, and its antagonists have been effectively used to prevent or reduce these symptoms. However, these antagonists often affect cardiac function, such as QT interval prolongation, limiting their daily use. The study aimed to explore the inhibitory effects of Panaxadiol on recombinant 5-HT3α receptors. The results indicated that Panaxadiol saponins (TS, PD, PT, Rb1, Rg1) exhibited considerable inhibitory effects on 5-HT3α receptors, suggesting that certain types of Panaxadiol saponins may possess anti-nausea and vomiting properties.
Non-alcoholic fatty liver disease (NAFLD) is a syndrome characterized by the accumulation of triglyceride-rich lipids in liver cells, leading to a series of pathological changes in the liver. Ai Mi and colleagues identified a bioactive component, Panaxadiol saponin (PDS-C), isolated from total ginseng saponins and formulated into capsules named Pai-neng-da (PND). The study confirmed that PDS-C has multiple effects in promoting energy metabolism, regulating immune function, and improving oxidative damage. Researchers comprehensively evaluated the impact of PDS-C on glucose and lipid metabolism, oxidative stress, and inflammation in NAFLD.
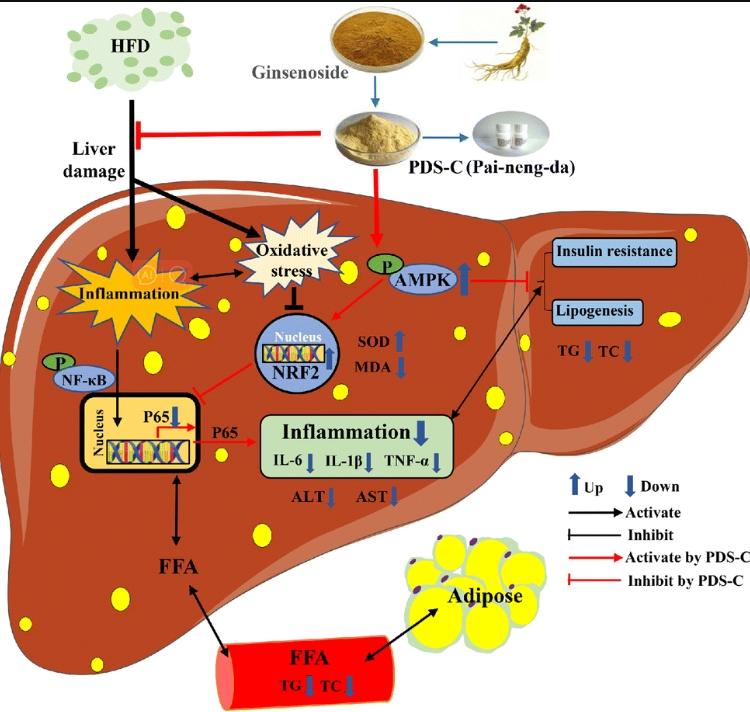
Research found that PDS-C exhibits a certain degree of glycemic control and anti-hepatic steatosis effects by activating AMPK, improving oxidative stress, and alleviating inflammatory damage. Our study suggests that PDS-C could be a potential hepatoprotective adjunct or dietary supplement for NAFLD, providing experimental evidence for future NAFLD treatments.
Studies have confirmed that PDS-C can effectively reverse cytopenia in mice and rats, and safely treat primary immune thrombocytopenia (ITP), aplastic anemia, bone marrow suppression, and cytopenia.
To achieve pharmacological effects with Panaxadiol, the dosage must be appropriate; too much or too little may not yield optimal results. For instance, Panaxadiol used to alleviate the side effects of acetic acid prednisolone should be dosed at 60 mg/kg for best results, as deviations in dosage can lead to less effective outcomes. Research on the pharmacology of individual ginseng components indicates that certain components may not produce side effects when used in appropriate amounts, and ginseng generally shows minimal side effects in therapeutic environments and doses, thus reducing the risk of adverse situations.
Panaxadiol is gaining attention for its potential health benefits, but its specific uses and effects may vary among individuals. Therefore, consulting a physician before using Panaxadiol for health improvement or therapeutic purposes is crucial. A physician can provide professional advice to ensure its use aligns with your health status, maximizing potential benefits while avoiding unnecessary risks and side effects.
[6]https://www.sciencedirect.com/topics/medicine-and-dentistry/panaxadiol
[7]Patel S, Rauf A. Adaptogenic herb ginseng (Panax) as medical food: Status quo and future prospects[J]. Biomedicine & pharmacotherapy, 2017, 85: 120-127.
[8]Tian S, Chen S, Feng Y, et al. Ginseng-derived panaxadiol ameliorates STZ-induced type 1 diabetes through inhibiting RORγ/IL-17A axis[J]. Acta Pharmacologica Sinica, 2023, 44(6): 1217-1226.
[9]Wang Z, Li M Y, Zhang Z H, et al. Panaxadiol inhibits programmed cell death-ligand 1 expression and tumour proliferation via hypoxia-inducible factor (HIF)-1α and STAT3 in human colon cancer cells[J]. Pharmacological Research, 2020, 155: 104727.
[10]Mi A, Hu Q, Liu Y, et al. Hepatoprotective efficacy and interventional mechanism of the panaxadiol saponin component in high-fat diet-induced NAFLD mice[J]. Food & Function, 2024, 15(2): 794-808.
[11]Xiao-Li Li, Chong-Zhi Wang, et al. (2009). Panaxadiol, a purified ginseng component, enhances the anti-cancer effects of 5-fluorouracil in human colorectal cancer cells. , 64(6), 1097–1104. doi:10.1007/s00280-009-0966-0
 |
 |