What is Sodium Acetate Trihydrate? Sodium acetate trihydrate, also known as trihydrate sodium acetate, is a chemical compound widely used in various industries. It serves as a preservative and acidulant in the food additive industry, such as in meat preservation and neutralizing sulfuric acid waste. With excellent antibacterial properties, it is also utilized as an acid wash agent in industrial sectors like photography and printing. In the environmental management industry, sodium acetate trihydrate is a valuable chemical reagent, particularly in wastewater treatment processes as an external carbon source, aiding in pH adjustment, precipitation, microbial oxidation, and other steps to facilitate sewage treatment. Additionally, it finds extensive applications in agriculture and agrochemistry, helping to neutralize acidic soils and fertilizers for better absorption. The main focus of this article is to understand the properties, chemical formula, and uses of sodium acetate trihydrate.
Sodium acetate trihydrate is a compound with a unique molecular structure and versatile applications. Composed of sodium, carbon, hydrogen, and oxygen atoms, its chemical formula is C6H9NaO6·3H2O. Sodium acetate trihydrate is formed from the reaction of acetic acid with sodium hydroxide. This reaction initially yields sodium acetate, which further reacts with acetic anhydride to produce sodium acetate trihydrate. When crystallized, it forms a white, odorless, water-soluble compound. Structurally, sodium acetate trihydrate is characterized by the combination of sodium ions with acetate groups, forming a stable molecular lattice. Each sodium acetate molecule contains three water molecules, resulting in its hydrated state.
One of its remarkable characteristics is its ability to undergo rapid phase transition between solid and liquid states. This property, known as supercooling, allows it to release heat upon crystallization, making it useful in hand warmers and reusable heat packs.
Sodium acetate trihydrate finds widespread applications across various fields including meat preservation, neutralizing sulfuric acid waste, acid wash agents, photoresists, and acid washing agents. Additionally, as a carbon source in wastewater treatment, it plays a crucial role in environmental management by helping to adjust sewage pH, precipitate impurities, and facilitate microbial oxidation processes. In agriculture, it can be used to neutralize acidic soils and fertilizers for improved absorption.
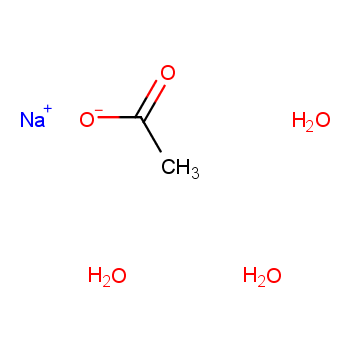
What is the formula for sodium acetate trihydrate? The chemical formula of sodium acetate trihydrate is NaCH3COO·3H2O, a compound consisting of sodium ions (Na+), acetate ions (CH3COO-), and water molecules. This formula indicates that for each sodium acetate molecule, there are three water molecules associated with it. Upon decomposition, sodium acetate trihydrate breaks down into its molecular components: sodium acetate and water. When heated, the trihydrate loses water, transforming into anhydrous sodium acetate and releasing water molecules. This decomposition process is reversible as anhydrous sodium acetate can reabsorb moisture from the atmosphere, reverting to its trihydrate form. This property makes it valuable in applications where controlled release of water is needed, such as hand warmers and heat packs. Additionally, sodium acetate is widely used in the food industry as a flavor enhancer and preservative, and in chemical laboratories as a buffering agent. Due to its stability and ease of handling, its trihydrate form is particularly useful in these applications.
Understanding the chemical formula of sodium acetate trihydrate is crucial for optimizing its applications across various fields, from chemistry to pharmaceuticals.
Sodium acetate trihydrate possesses several notable physical and chemical properties. At room temperature, it exists as colorless transparent crystals. What is the melting point of sodium acetate trihydrate? One of the most significant characteristics of sodium acetate trihydrate is its relatively low melting point, approximately 58 degrees Celsius, accompanied by a phenomenon known as "heat of crystallization." This property makes sodium acetate trihydrate valuable in heat packs and hand warmers as it undergoes rapid exothermic crystallization upon triggering, releasing stored heat. The melting point of phase change material sodium acetate trihydrate is around 58°C, with a melting enthalpy of approximately 265 kJ/kg, making it a promising medium-temperature phase change energy storage material. Additionally, it exhibits high solubility in water, facilitating dissolution and preparation of solutions suitable for various applications in chemical laboratories and industrial processes. Its aqueous solution possesses buffering capacity, making it highly useful in maintaining pH levels in chemical reactions and biochemical analyses.
Sodium acetate trihydrate is relatively stable under normal conditions and can have an extended shelf life with proper storage, making it a reliable reagent in laboratory environments. Its low melting point and heat release upon crystallization make it a preferred choice for reusable heat packs, providing cost-effective and convenient solutions for therapeutic heating needs. Its high solubility and buffering capacity make it an essential component in various chemical processes such as dyeing textiles, synthesizing pharmaceuticals, and adjusting pH in biochemical analyses. The stability of sodium acetate trihydrate ensures consistent performance and reliable results, contributing to its widespread use in research laboratories and manufacturing facilities.
The production and manufacturing process of sodium acetate trihydrate typically involve the following steps: Firstly, acetic acid is added to a reaction vessel, and sodium hydroxide solution is added for neutralization, with the resulting carbon dioxide gas being neutralized. Then, after filtration, the reaction product undergoes evaporation, crystallization, and centrifugal drying to obtain the final product of sodium acetate trihydrate solid, with water molecules incorporated into the lattice structure.
The purity and quality of raw materials, as well as precise control over reaction conditions such as temperature and pressure, play a crucial role in determining the yield and properties of the final product. The manufacturing process of sodium acetate trihydrate is essential to ensure product quality and consistency. Any deviations in synthetic methods or impurities in raw materials can significantly affect the properties and performance of the final product. For example, impurities introduced during the synthesis process can alter the chemical composition or crystal structure, affecting its solubility, stability, and reactivity. Therefore, strict quality control measures are implemented at each stage of the production process to maintain high standards and meet the stringent requirements of various industries. By understanding and optimizing the manufacturing process, manufacturers can tailor the performance of sodium acetate trihydrate to suit specific applications, thereby contributing to its extensive use in different fields.
Here is an example of using acetic acid from PTA oxidation residue to produce sodium acetate trihydrate: 1000L of mother liquor, recovered from the PTA oxidation residue recovery workshop, is introduced into a distillation kettle for distillation under stirring and reduced pressure to distill out benzoic acid. The stirring speed is 70 rpm, the vacuum degree is -0.085, and the kettle temperature is 65°C; the purified glacial acetic acid is added into the reaction kettle and slowly added with flake sodium hydroxide under stirring and water cooling conditions. The stirring speed is 70 rpm, the reaction temperature is 65°C, and the end point pH is controlled to be 11. After adding sodium hydroxide, it is kept warm for 1 hour; the reaction solution of sodium acetate obtained after neutralization is poured into a distillation kettle for heating and evaporation under stirring and reduced pressure. The stirring speed is 70 rpm, the vacuum degree is -0.075 MPa, and the kettle temperature is 75°C. When the concentration of sodium acetate reaches 80%, the pH of the solution is measured. When the pH value exceeds 9, the reaction solution of sodium acetate is acidified until the final pH value is 8; the evaporated and concentrated reaction solution is cooled to 75°C, and then poured into a filter for filtration. The filtrate enters the reaction kettle, and the filter cake is sodium benzoate; the filtrate in the reaction kettle is stirred and cooled. When the temperature drops to 55°C, 1% of the theoretical amount of sodium acetate is added to the filtrate as a crystallization inducer. The cooling time is controlled for 4.5 hours, and then cooled to room temperature. It is poured into a filter until dry. The filtered mother liquor is returned to the distillation kettle for reuse, and the filter cake is wet sodium acetate trihydrate; the wet sodium acetate trihydrate is placed in a vacuum dryer for vacuum drying. The vacuum degree is -0.087 MPa, and the temperature is 38°C. After drying to a constant, the product is obtained.
Sodium acetate trihydrate is a chemical widely used in various industries, playing an indispensable role in chemical experiments, medicine, and the food industry. In chemical experiments, it is used as a buffering agent and metal surface cleaning agent; in the food industry, it is a common food additive used to enhance the taste and flavor of food, such as in fireworks, dyes, food additives, and detergents production; in the medical field, sodium acetate trihydrate is widely used in the production of anticoagulants, as a pharmaceutical intermediate, and to improve drug quality.
In medical applications, sodium acetate trihydrate is an important raw material for drug production. For example, it can be used in the production of anticoagulants, improving drug quality, and supporting the development and innovation of the pharmaceutical industry. At the same time, sodium acetate trihydrate plays an important role in controlling the pH rise of denitrifying sludge and reducing the effluent COD value.
Sodium acetate trihydrate is also used in meat preservation, textile industry, neutralizing sulfuric acid waste, as an acid wash agent in chrome tanning, delaying the sulfuration of chloroprene during the production of synthetic rubber, etc. Additionally, sodium acetate trihydrate can serve as a supplementary carbon source, controlling the rise of pH in denitrifying sludge while reducing the effluent COD value.
Sodium acetate trihydrate is a corrosive and irritant substance. Therefore, proper gloves and goggles must be worn during handling or storage. Direct contact with the skin or eyes can cause severe discomfort and pain. Sodium acetate trihydrate is hygroscopic, and dust can cause it to crystallize, so special attention should be paid to prevent dust from entering the atmosphere to avoid respiratory problems.
Although sodium acetate trihydrate has widespread applications in the chemical industry, it also poses some environmental and ecological issues. Its use as a carbon source in wastewater treatment is crucial, but if not handled properly, it may pose potential hazards to water bodies and the environment. Therefore, appropriate environmental protection measures must be taken when handling or storing sodium acetate trihydrate, such as adopting advanced wastewater treatment technologies and controlling waste discharge, to ensure environmental safety and sustainable development.
Sodium acetate trihydrate is an important compound with various applications in many industries. Its synthesis involves the combination of acetic acid and sodium hydroxide, resulting in a crystalline form containing water molecules. While it is significant in food preservation, chemical synthesis, and heating pads, it is essential to recognize potential hazards associated with its handling, including irritation upon contact with skin or eyes. By taking appropriate precautions, its multifunctionality proves invaluable across various fields. I encourage readers to delve into the uses and precautionary measures of sodium acetate trihydrate to effectively harness its potential while ensuring safe practices in handling and utilization.
[1] https://go.drugbank.com/salts/DBS
[2] Liaoyang Hongwei District Synthetic Catalyst Factory. Method for Producing Sodium Acetate Trihydrate from PTA Oxidation Residue. 2010-06-23.
[3] Fang Guihua, Zhang Wentao, Yu Menghuan, et al. Research Progress on Phase Change Energy Storage Material Sodium Acetate Trihydrate [J]. New Chemical Materials, 2021, 49(08):16-20. DOI:10.19817/j.cnki.issn1006-3536.2021.08.004.
 |
 |
 |