
Lewis structures, devised by Gilbert N. Lewis, visually represent electron arrangements in molecules. By depicting valence electrons as dots and bonds as lines, Lewis structures predict a molecule's shape and properties based on the octet rule. This rule states that atoms tend to achieve stability by having eight electrons in their outer shell. Lewis structures adhere to this rule, offering a clear picture of chemical bonding.
Iodine dioxide (IO2) is a compound consisting of one iodine atom bonded to two oxygen atoms. It is typically used in various industrial applications and chemical research. Despite being less common than other iodine compounds, IO2 plays a significant role in certain chemical processes and reactions.
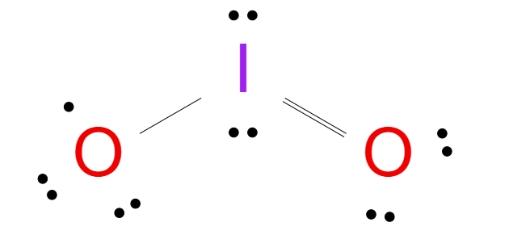
Let's dive into drawing the Lewis structure of IO2:
Step 1: Identify the Central Atom: Iodine (I) is the central atom in IO2 because it's less electronegative than oxygen.
Step 2: Calculate Total Valence Electrons: Iodine contributes 7 valence electrons, and each oxygen contributes 6, giving a total of 7 + (2 x 6) = 19 valence electrons.
Step 3: Arrange Electrons Around Atoms: Connect each oxygen atom to the central iodine atom with a double bond (two lines) and distribute the remaining electrons as lone pairs around each oxygen atom.
Step 4: Fulfill the Octet Rule: Ensure each oxygen atom has 8 electrons (2 lone pairs and 2 bonding pairs), and the iodine atom has 8 electrons (2 lone pairs and 4 bonding pairs).
Step 5: Check for Formal Charges: Formal charges may not be necessary as all atoms have achieved the octet rule.
The structure of Iodine dioxide comprises a central Iodine atom around which 18 electrons or 9 electron pairs are present, including one lone pair on the iodine atom. Therefore, the molecular geometry of IO2 will be bent or V-shaped.
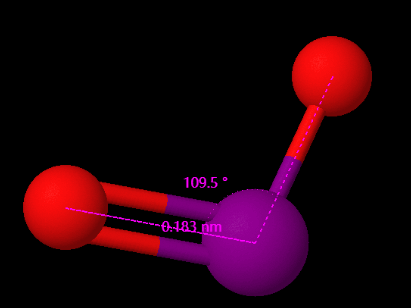
This theory addresses electron repulsion and the need for compounds to adopt stable forms. In IO2, two pi bonds form between iodine and oxygen, with lone pairs on each oxygen atom. Although iodine has only four valence orbitals, the Lewis structure suggests two bond pairs and one lone pair, implying the use of p-orbitals in this complex. Advanced calculations reveal the electronic structure consists of two delocalized bonds across all three atoms, rather than distinct bonds involving d-orbitals.
The Lewis structure suggests that IO2 adopts a bent or V-shaped geometry. In this arrangement, the two oxygen atoms are symmetrically positioned around the central iodine atom, forming two bond pairs. This geometry minimizes electron-electron repulsion, resulting in a stable configuration.
The orbitals involved and the bonds produced during the interaction of Iodine and oxygen molecules will be examined to determine the hybridization of Iodine dioxide. 5s, 5px, 5py, 5pz, and 5dxy are the orbitals involved. The Iodine atom, which is the central atom in its ground state, will have the 5s25p5 configuration in its formation.
The electron pairs in the 5s and 5px orbitals become unpaired in the excited state, and one of each pair is promoted to the unoccupied 5dxy orbital. All five half-filled orbitals (one 5s, three 5p, and one 5d) hybridize now, resulting in the production of three sp3d hybrid orbitals.
The bond angle in IO2 is approximately 109.5 degrees. This angle arises from the bent geometry of the molecule, where the two oxygen atoms are positioned at an angle to the central iodine atom, resulting in a bond angle of approximately 109.5 degrees between adjacent oxygen atoms. The bond length in IO2 is approximately 183 pm.
| Iodine Dioxide Cas 13494-92-3 | |
| Molecular formula | IO2 |
| Molecular shape | Bent (V-shaped) |
| Polarity | Polar |
| Hybridization | sp3d hybridization |
| Bond Angle | Approximately 109.5 degrees |
| Bond length | Approximately 183 pm |
To determine if a Lewis structure is polar, examine the molecular geometry and bond polarity. In the case of iodine dioxide (IO2), the Lewis structure shows iodine at the center bonded to two oxygen atoms. IO2 has a bent geometry, where the two oxygen atoms are asymmetrically arranged around the iodine atom. Although the I-O bonds are polar, the asymmetry of the molecule causes the dipole moments not to cancel out, making IO2 a polar molecule.
To calculate the total bond energy of IO2, first, look up the bond energy for a single iodine-oxygen (I-O) bond, which is approximately 200 kJ/mol. IO2 has two I-O bonds, so you multiply the bond energy of one I-O bond by the number of bonds. This gives a total bond energy of 400 kJ/mol for IO2. This value represents the energy required to break all the I-O bonds in one mole of IO2 molecules.
Bond order is the number of chemical bonds between a pair of atoms. In the Lewis structure of IO2, each iodine-oxygen bond is a double bond, so the bond order for each I-O bond is 2. If a molecule has resonance structures, bond order is averaged over the different structures, but IO2 does not have resonance, so the bond order remains 2.
Electron groups in a Lewis structure include both bonding pairs (shared electrons) and lone pairs (non-bonded electrons) around an atom. In IO2, each iodine atom has three electron groups around it, corresponding to the two I-O bonds (two bonding pairs and one lone pair on iodine).
In a Lewis dot structure, the dots represent valence electrons. Each dot corresponds to one valence electron of an atom. In IO2, iodine is surrounded by two bonding pairs (represented by lines in the Lewis structure) and one lone pair (represented by two dots). The dots help visualize how electrons are shared or paired between atoms.
 |
 |