The film "Cleopatra," produced by Fox and ultimately winning multiple Oscar awards, tells the legendary life of the Egyptian Queen Cleopatra. She formed political alliances with Julius Caesar and Mark Antony, which successfully delayed the Roman annexation of Egypt, allowing the kingdom to maintain relative independence amidst Rome's expansion and providing a strategic buffer period. The film's dazzling golden serpent jewelry, the tragic end of the queen with a snake bite, and Elizabeth Taylor’s portrayal of a "femme fatale" all highlight the unique symbolism of snakes in Egyptian culture.

In ancient Rome, snakes also had rich symbolic meanings, one of which was their representation of healing and redemption in medicine. Asclepius, the god of medicine and the son of Apollo, is often depicted holding a snake-entwined staff. In modern medicine, the symbol of the caduceus (the snake-entwined staff) is still widely used, such as in the logo of the World Health Organization. Additionally, the Bowl of Hygieia, the cup held by the goddess of cleanliness and preventive medicine, features a snake coiled around it, symbolizing one of the most widely recognized international symbols in pharmacology.

Regarding the medicinal use of snakes, many people first heard of it from martial arts novels, and later learned from the work of Liu Tsung-yüan, "Catching Snakes": “In the wilds of Yongzhou, there is a strange snake, black in color with white markings, which kills plants and trees it touches; when it bites a person, there is no cure. However, when dried and used as bait, it can cure conditions like wind-related ailments, muscle spasms, and infections...”. This concept of using snakes in medicine became more concrete for me.
I then shared with my friends the story of a certain drug, which also has a connection to venomous snakes.
Hypertension has always been a difficult problem to solve in the medical field. Research into its mechanisms dates back more than a century.
In 1896, two researchers from Sweden conducted a fascinating experiment. They injected kidney extracts into rabbit veins and discovered that the extracts could rapidly raise the rabbit’s blood pressure. From this, they concluded that the kidneys secreted a hormone that could promote blood vessel constriction and speculated that this hormone might be linked to kidney disease and hypertension. They named this hormone "renin."
For many years, their hypothesis was largely ignored, until mid-20th century scientists confirmed their idea. Researchers found that renin did not directly affect blood vessels but instead triggered the conversion of Angiotensin I (Ang I), which had no hypertensive effect, into the powerful vasoconstrictor Angiotensin II (Ang II) via the Angiotensin-Converting Enzyme (ACE). Ang II not only causes blood vessels to constrict but also increases aldosterone levels, forming the renin-angiotensin-aldosterone system (RAAS).
Imbalances in this system can lead to hypertension and cardiovascular complications such as strokes, heart attacks, and heart failure. This gave researchers a new avenue to explore—substances that could inhibit or regulate the RAAS system could potentially treat hypertension and its complications.

In 1933, Silva, a medical student from the University of S?o Paulo, Brazil, witnessed a patient who died from a Brazilian pit viper bite. During treatment, he observed that despite all efforts, the patient’s blood pressure could not be raised during shock. This mysterious event stayed in Silva’s mind, and he began to wonder: Why couldn’t the patient’s blood pressure rise?
Could there be something mysterious in the snake venom? With this question in mind, Silva eventually succeeded in isolating a special peptide from snake venom that caused blood vessel dilation and named it "bradykinin." However, this peptide had a very short half-life in the bloodstream and became ineffective within minutes, rendering it largely impractical.
In 1965, Silva’s student Ferreira discovered that snake venom could enhance the action of bradykinin, so he speculated that there might be another substance in the venom that could inhibit bradykinin degradation. After various attempts, he successfully isolated this substance, naming it the "bradykinin potentiating factor (BPF)." Unfortunately, BPF also had a very short lifespan in the human body.
Years later, Ferreira went to London to work under Professor Vane, who won the Nobel Prize in Physiology or Medicine in 1982. Professor Vane suggested that Ferreira study the effect of BPF on the renin-angiotensin system. Reluctantly, Ferreira agreed. After further research, Ferreira and his colleagues discovered that BPF had two functions: it enhanced bradykinin’s effects and inhibited the conversion of Ang I to Ang II. This led Professor Vane to realize that isolating and purifying ACE inhibitors (ACEIs) from snake venom could be an effective approach at the time.
As an advisor to the American laboratory of Schering-Plough (the predecessor of Bristol-Myers Squibb), Professor Vane immediately recommended that the research department focus on studying snake venom extracts to develop ACE inhibitors. With funding from Schering-Plough, protein chemist Professor Andretti and enzymologist Professor Kusman embarked on a journey to develop ACE inhibitors.
Over the next two years, these talented scientists isolated and synthesized various peptide compounds from snake venom, and pharmacological experiments showed that several of them effectively inhibited ACE. One compound, which could inhibit ACE activity for a longer period, was named teprotide. They synthesized 1 kilogram of it for clinical research.
Unfortunately, the U.S. Food and Drug Administration (FDA) rejected teprotide’s application for clinical trials. Professor Vane had to take teprotide to the UK for clinical research, which proved successful, showing that teprotide could effectively inhibit Ang I and reduce blood pressure. However, teprotide had a significant drawback: as a large peptide compound, it could not be absorbed orally and could only be administered by injection, limiting its clinical application.
Given this, Professors Andretti and Kusman shifted their focus to developing a small-molecule ACEI that could be absorbed orally. However, due to the lack of knowledge about ACE’s structure at the time, progress was slow. As if things couldn’t get worse, Professor Vane suddenly left the project, and Schering-Plough had to pause the research.

Although Schering-Plough no longer supported the project, Professors Andretti and Kusman persisted with their research, believing that success was within reach.
In 1974, Professor Kusman came across an article about a new carboxypeptidase A inhibitor called benzylsuccinic acid. He quickly called Professor Andretti to discuss it. They speculated that the active site of ACE might be similar to that of carboxypeptidase A and decided to use the known structure of carboxypeptidase A to guide the development of ACEIs. This insight led them to propose the new concept of "structure-based drug design." With a clear new direction, the two scientists began modifying and optimizing the compound structure.
After dozens of iterations, they successfully designed and synthesized a new compound that inhibited ACE activity thousands of times more effectively and could be absorbed orally. They named this compound Captopril, a new class of ACE inhibitors was born! The concept of "structure-based drug design" became a fundamental strategy for the development of new drugs worldwide and laid the foundation for modern computer-aided drug design.
In 1977, Captopril underwent extensive clinical trials and was approved by the FDA four years later as the world’s first ACE inhibitor on the market. It also became Schering-Plough’s first product to achieve over $1 billion in annual sales. In addition to its blood pressure-lowering effects, Captopril also brought unexpected benefits in treating heart failure, significantly reducing mortality rates in affected patients. As a result, Professors Andretti and Kusman were awarded the Lasker Clinical Medical Research Award in 1999, one of the highest honors in clinical medicine, second only to the Nobel Prize in Physiology or Medicine.
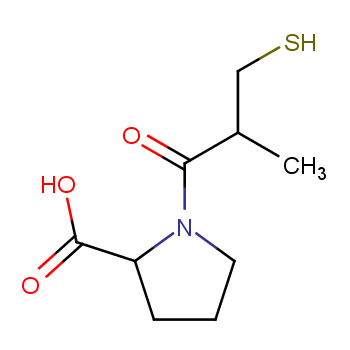

The development of Captopril is a classic case of "from natural observation to mechanism exploration to drug design," showcasing the close integration of basic research and translational medicine. Its creation not only saved countless lives of hypertensive and heart failure patients but also paved the way for the development and application of ACE inhibitors, guiding the research of subsequent new ACEI drugs.
 |
 |