Acetic acid (CH3COOH), also known as ethanoic acid, is a weak acid that can partially dissociate into acetate ions and hydrogen ions in aqueous solutions. It is a colorless and transparent liquid at room temperature. In the pharmaceutical field, acetic acid is a commonly used organic reagent, such as in the preparation of buffer solutions and pH adjustment of solutions. However, very few formulation researchers are aware of the ingenious applications of acetic acid in pharmaceutical formulation development. It is not an exaggeration to say that acetic acid, a seemingly insignificant chemical reagent, can revive some nearly “dead” formulation development projects, significantly advancing the project timeline.
For certain poorly soluble drugs, preparing cyclodextrin complexes is an effective formulation method to enhance their solubility. However, those who have worked on such projects know that drugs often cannot be completely dissolved in cyclodextrin solutions. This frequently leads to a reduction in the drug load, which may result in an oversized final formulation (e.g., tablet volume). Additionally, cyclodextrin complexation efficiency (CE) is often less than ideal, leading to complexes containing significant amounts of unencapsulated drug, with only a portion of the cyclodextrin molecules truly forming complexes with the drug.
Some researchers may attempt to add organic solvents (e.g., ethanol). While this can increase the apparent intrinsic solubility (S0) of the drug and promote the shift of the complexation equilibrium towards complex formation, organic solvent molecules will compete with the drug for the cyclodextrin cavity, lowering the apparent stability constant (Kc) of the drug/cyclodextrin complex and ultimately decreasing the overall complexation efficiency.
For certain basic drugs, acetic acid serves as an excellent "ingredient" in cyclodextrin complex formation (for acidic drugs, ammonia water is typically used, but since this article focuses on acetic acid, ammonia water is not discussed here). The principle is that when a volatile weak acid (such as acetic acid) is added to a slightly basic drug in an aqueous complexation system, or a volatile weak base (such as ammonia water) is added to a slightly acidic drug system, the acid-base reaction promotes drug ionization. After ionization, the apparent solubility (S0) of the drug increases, and more drug molecules can dissolve in the solution. According to the principle of complexation equilibrium, an increase in S0 (which increases the reactant concentration) shifts the equilibrium towards the formation of the drug-cyclodextrin complex, thus temporarily improving complexation efficiency. After complexation is complete, volatile acids or bases (such as acetic acid or ammonia water) can be removed through drying (e.g., vacuum drying). At this point, the drug reverts to its non-ionized form, resulting in a non-ionized drug/cyclodextrin complex.
AR-A000002 is a selective serotonin receptor antagonist developed by AstraZeneca. It is a weakly basic drug (pKa=8.3) with the chemical structure shown in Figure 1.

The drug has poor water solubility, and its cyclodextrin complex was prepared by complexing AR-A000002 with equimolar amounts of hydroxypropyl-β-cyclodextrin (HP-β-CD), with the addition of 20 equivalents of acetic acid to promote drug dissolution. After the drug dissolved, the complexation solution was freeze-dried to obtain the drug complex. After freeze-drying, the molar ratio of drug, cyclodextrin, and acetic acid was approximately 1:1:1. After heating in a vacuum oven at 88°C for 7 hours, the acetic acid content dropped to around 0.1 equivalent. The heating was continued for 20 days, and the residual acetic acid content gradually decreased to 0.08 equivalent.

The dissolution test results show that the AR-A000002/HP-β-CD complex prepared with acetic acid dissolved rapidly (black circles in Figure 2), forming a supersaturated solution, releasing about 45% of the drug. This supersaturated solution remained stable for about 10 hours before precipitation. In contrast, the complex prepared without acetic acid did not form a supersaturated solution (white circles in Figure 2), and the drug AR-A000002 (white square in Figure 2) dissolved very slowly.
I have participated in several spray-dried solid dispersion projects, and one of the most troublesome issues is the poor solubility of many new drugs. During early solvent screening, some drugs have very low solubility in common organic solvents (e.g., ethanol, methanol, acetone, dichloromethane, tetrahydrofuran, methanol/dichloromethane mixed solvents), which leads to the need for large amounts of organic solvents during scale-up production, restricting production efficiency and increasing production costs.
For weakly basic drugs, volatile acids can be used to improve drug solubility in spray-drying solvents by ionization. After vacuum drying, the volatile acids evaporate, allowing the drug to revert to its free form. Acetic acid is an ideal volatile acid because its pKa is moderate (4.75), and compared to strong acids like hydrochloric acid, it does not form tight complexes with weakly basic drugs. Strong acids such as hydrochloric acid are unlikely to dissociate from basic drugs due to the strength of the chemical bonds, while acetic acid can evaporate during subsequent vacuum drying, allowing the drug to revert to its free base form.
Researchers at Lonza discovered that for the weakly basic drug gefitinib (pKa=7.2, chemical structure shown in Figure 3), acetic acid could increase its solubility in the MeOH:H2O spray-drying solvent by 10 times (using acetic acid at 20mg/mL) by forming transient acetate salts.
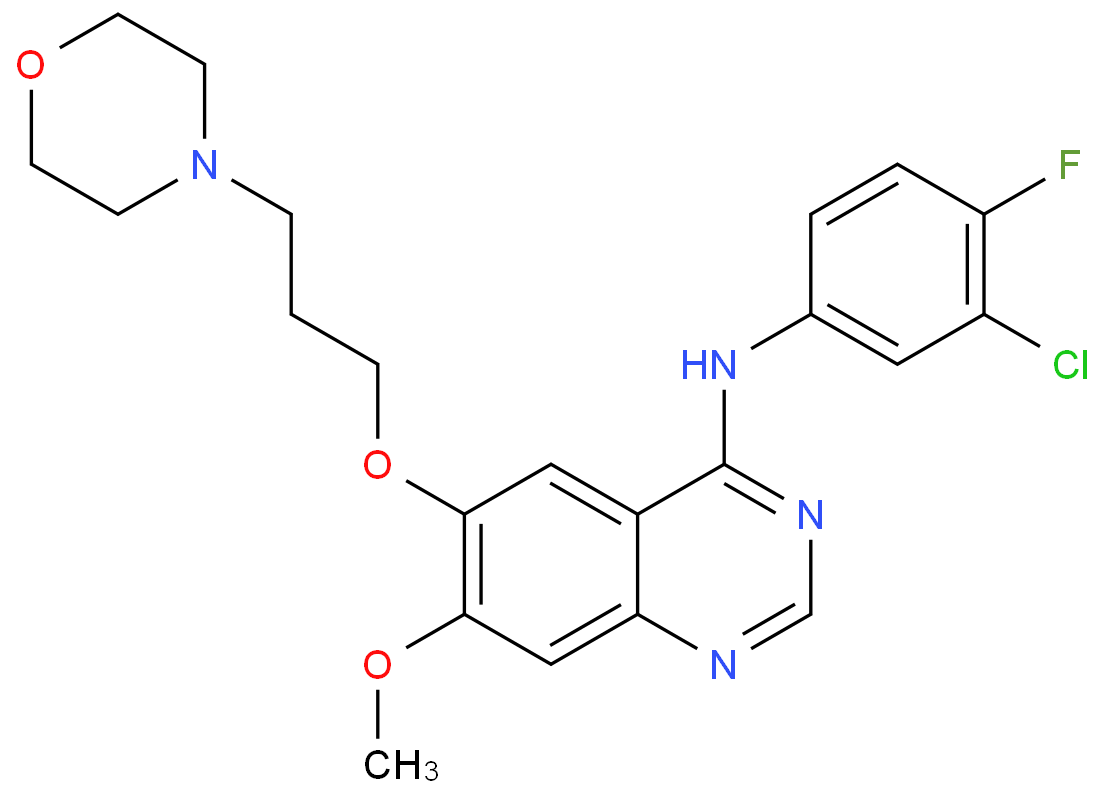
Acetic acid is removed during the subsequent vacuum drying process, and the resulting spray-dried dispersion of gefitinib shows similar properties to the dispersion prepared without acetic acid, including morphology (observed by SEM), amorphous state (detected by PXRD), glass transition temperature (measured by mDSC), and in vitro dissolution, indicating no significant difference in the characteristics of gefitinib. This suggests that the use of acetic acid does not significantly affect the performance of gefitinib in the spray-dried dispersion, and it is expected to perform similarly in vivo.
Therefore, we can see that simply adding acetic acid to the spray-drying solvent significantly improves the drug's solubility, which not only increases production efficiency and reduces the amount of solvent required, making the large-scale production of poorly soluble drugs feasible but also contributes to sustainable green development and environmental protection.
 |
 |
 |