Calcium hypochlorite is a commonly used bleaching and disinfecting agent, primarily composed of calcium chloride and calcium hypochlorite. In bleaching, the primary role of calcium hypochlorite is to destroy the 'chromophore' of organic pigments through the strong oxidative properties of hypochlorite, irreversibly achieving the bleaching effect. By reading this article, readers will gain a deep understanding of calcium hypochlorite bleaching powder and its various applications, ensuring safe and effective usage in their respective industries.
Calcium hypochlorite bleaching powder is a strong oxidizing agent, primarily composed of calcium hypochlorite. It is a white or grayish-white powder that exhibits strong oxidative properties upon contact with water, swiftly removing color or pigments. Widely used in food and textile processing, as well as in the field of disinfection, calcium hypochlorite bleaching powder differs from ordinary bleaching powder in its chemical composition and properties. While bleaching powder typically refers to a mixture containing calcium hypochlorite, calcium hypochlorite bleaching powder consists solely of calcium hypochlorite. The primary components of bleaching powder may also include calcium chloride, sodium chloride, and calcium hydroxide.

(1) Detailed Breakdown of Calcium Hypochlorite Chemical Structure
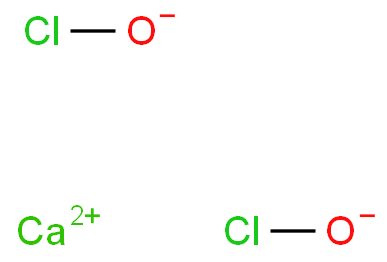
The chemical formula of calcium hypochlorite is Ca(ClO)2, which undergoes a complex decomposition process upon dissolution in water, generating hypochlorous acid (HOCl) and calcium hydroxide (Ca(OH)2). This decomposition occurs by releasing chlorine gas and oxygen, forming these by-products. The produced hypochlorous acid is a potent oxidizing agent crucial for its bleaching action. Through a series of chemical reactions, hypochlorous acid attacks the pigments in stains, disrupting their molecular structures and rendering them colorless. Meanwhile, calcium hydroxide helps maintain the alkaline pH environment necessary for effective bleaching.
(2) Explanation of Bleaching Mechanism
The bleaching mechanism of calcium hypochlorite involves its ability to oxidize chromophores, which are responsible for coloring organic compounds. When calcium hypochlorite is applied to a dyed surface, the generated hypochlorous acid reacts with the chromophores, altering their chemical structures or breaking them down into smaller colorless molecules, thus removing their color effectively. This process efficiently eliminates visible stains, leaving a cleaner and brighter surface. Additionally, the alkaline environment produced by calcium hydroxide aids in the penetration of hypochlorite into fabrics or surfaces, enhancing the bleaching action and ensuring thorough stain removal.
(1) Application in Water Treatment Processes
Due to its potent bleaching and disinfecting properties, calcium hypochlorite finds extensive applications across various industries. In water treatment processes, calcium hypochlorite plays a crucial role in purifying drinking water and wastewater. When added to water, it releases hypochlorous acid, a powerful oxidizing agent capable of effectively eliminating harmful microorganisms, including bacteria, viruses, and algae. This disinfection process helps ensure the safety and potability of drinking water and other household water uses. Additionally, calcium hypochlorite aids in the removal of organic impurities and unpleasant odors, further improving water quality.
(2) Whitening Fabrics in Textile Industry
In the textile industry, calcium hypochlorite is used for whitening fabrics. Fabrics undergo bleaching to remove natural pigments, stains, and impurities, achieving the desired pristine white appearance for various end products. As a bleaching agent, calcium hypochlorite oxidizes the chromophores in fabrics, thereby disrupting their molecular structures and rendering them colorless. This results in brighter and whiter fabrics, aesthetically pleasing and suitable for dyeing or further processing. Moreover, the use of calcium hypochlorite ensures effective and uniform bleaching of different types of textiles, from cotton to synthetic fibers.
(3) What is calcium oxychloride bleaching powder used for?
In addition to industrial applications, calcium hypochlorite is a key ingredient in many household cleaning products, including bleaches and disinfectants. These products leverage the potent oxidizing properties of calcium hypochlorite to remove stubborn stains, disinfect surfaces, and eliminate harmful bacteria and germs. Whether used in laundry detergents to brighten clothes or in surface cleaners to disinfect kitchen and bathroom surfaces, calcium hypochlorite provides effective cleaning and disinfection, contributing to hygienic and healthy living environments. However, caution must be exercised in the use of calcium hypochlorite products, following recommended dilution ratios and safety guidelines to prevent skin irritation or surface damage.
(1) Advantages over Other Bleaching Agents
As a widely used disinfectant, calcium hypochlorite offers high efficiency and safety, coupled with ease of use, making it prevalent for disinfection in households, businesses, and public spaces. The benefits of calcium hypochlorite over other bleaching agents lie in its high efficacy and safety. Compared to other bleaching agents, calcium hypochlorite exhibits higher bleaching effectiveness and lower toxicity, posing minimal harm to human health. Furthermore, calcium hypochlorite possesses a certain tolerance and antibacterial properties, capable of eradicating various bacteria and viruses at certain concentrations, thus safeguarding public health.
(2) Potential Drawbacks
However, calcium hypochlorite also presents some potential drawbacks and limitations in its usage: (1) Improper use may lead to the generation of toxic gases and hazardous substances, causing harm to human health. (2) Residues of calcium hypochlorite may have environmental impacts, potentially contaminating water sources and soil if mishandled. (3) The production process of calcium hypochlorite may contribute to pollution; for instance, in the production processes of calcium hypochlorite via the calcium method and the sodium method, issues such as calcium method overcapacity and sodium method still having room for growth need attention and resolution.
(1) Different Applications (Water Treatment, Cleaning, etc.)
To effectively utilize calcium hypochlorite powder, it is crucial to follow step-by-step instructions for its preparation and application. Firstly, determine the required concentration of hypochlorite solution based on the intended use (water treatment, cleaning, or disinfection). Measure the appropriate amount of calcium hypochlorite powder using a scale or measuring spoon, and dissolve it in the appropriate volume of water, preferably in a well-ventilated area to avoid inhalation of chlorine gas. Thoroughly stir the solution until the powder is completely dissolved, ensuring uniform concentration.
(2) Safety Precautions and Proper Handling Techniques
When preparing calcium hypochlorite solution for water treatment, use the recommended dosage based on the volume and quality of water to be treated. Generally, chlorine concentrations ranging from one to three parts per million (ppm) are sufficient for disinfection purposes, while shock chlorination or algae control may require higher concentrations. For household or industrial cleaning and disinfection purposes, follow the manufacturer's recommendations for dilution ratios and contact times to achieve effective results without compromising safety. When handling calcium hypochlorite powder or its solution, wear appropriate personal protective equipment such as gloves and goggles, avoid mixing with other chemicals to prevent hazardous reactions. Additionally, store calcium hypochlorite in a cool, dry place away from direct sunlight and moisture to maintain its stability and efficacy.
(1) Potential Hazards and Risks Associated with Use
Calcium hypochlorite is classified as a Class 5.1 dangerous goods under the International Maritime Dangerous Goods Code, as well as falling under Class 8 corrosive substances. Proper storage of calcium hypochlorite is essential in specialized hazardous goods warehouses, and it should be shipped according to formal hazardous goods declaration to prevent accidents such as fires or explosions. Moreover, calcium hypochlorite poses significant hazards to human health, with substantial vapor emissions being carcinogenic; therefore, caution is required during usage, especially during prolonged exposure or handling, necessitating the use of protective eyewear, gloves, and other safety gear.
(2) Recommended Protective Gear for Handling
When transporting calcium hypochlorite, safety precautions must be observed. For instance, professional ear protectors and dust masks should be used to prevent irritation from volatile gases and dust. Calcium hypochlorite export shipments require refrigerated transport, with a net weight per container not exceeding 14 tons, to prevent hazards caused by cargo leakage or impact.
(3) Safety Precautions and Proper Handling Techniques
Safety operation techniques for calcium hypochlorite also require significant attention. For example, before use, it must be ensured that the liquid inside the container has dried to prevent corrosion or explosion accidents. Meanwhile, when using calcium hypochlorite, avoid mixing with acids, alkalis, or other substances to prevent hazardous chemical reactions.
The use of calcium hypochlorite has certain environmental impacts, primarily manifested in water and soil pollution, as this chemical substance decomposes chlorine gas during the disinfection process, which is highly toxic to humans and other animals. In comparison with other bleaching methods, calcium hypochlorite does not hold superiority in sustainability. In contrast, photo-bleaching methods such as solar bleaching excel in sustainability, as they utilize renewable energy sources. Additionally, traditional bleaching technologies represented by calcium method calcium hypochlorite exhibit more significant negative environmental impacts, while although sodium method calcium hypochlorite applications have room for growth in environmental protection and sustainable development.
To mitigate the environmental impact of calcium hypochlorite usage, it is necessary to implement effective strategies for reducing environmental footprint, such as adopting environmentally friendly production methods, employing biotechnology, reducing chemical waste emissions, strengthening supervision and management of disinfection processes, among others. This not only alleviates environmental pollution issues but also promotes the stable development of the calcium hypochlorite market.
(1) Is calcium hypochlorite same as bleaching powder?
It is essential to clarify that although calcium hypochlorite is commonly referred to as a bleaching agent, it is not identical to household bleaching powder, which typically contains sodium hypochlorite. Calcium hypochlorite is a solid compound primarily used for bleaching and disinfection purposes, prevalent in industrial and water treatment environments. Understanding this distinction prevents confusion and ensures the accurate application of the appropriate product for specific tasks.
(2) What is calcium chloride bleaching powder used for?
Calcium hypochlorite powder is an effective disinfectant capable of eliminating harmful microorganisms in water sources, crucial for ensuring the safety and potability of drinking water.
(3) What is the use of calcium hypochlorite bleaching powder, and how does it compare to calcium hypochlorite bleaching powder?
Calcium hypochlorite bleaching powder is used as a disinfectant in aquaculture, with its primary component being calcium hypochlorite, and an effective chlorine content of 25-30%. It plays a vital role in pond disinfection and preventing bacterial diseases in fish, shrimp, and crabs. It can kill pathogenic microorganisms, preventing the occurrence of fish diseases. Additionally, it can oxidize pond sludge and water bodies, improving water quality. Its application involves dissolving and filtering after application, adhering to treatment courses, adjusting water quality, etc. Compared to calcium hypochlorite bleaching powder, calcium hypochlorite bleaching powder contains less calcium hypochlorite, hence does not require overly cautious protection of the skin, metal objects, and clothing during usage, to avoid problems caused by corrosiveness.
Calcium hypochlorite is a versatile and indispensable compound with wide-ranging applications in water treatment, textile bleaching, and household cleaning. Its potent bleaching and disinfecting properties make it a critical component for ensuring the safety and cleanliness of drinking water, while also enhancing the aesthetic appeal and quality of textiles in the textile industry. Moreover, its efficacy in stain removal and surface disinfection underscores its importance in household cleaning products. By harnessing the power of calcium hypochlorite, individuals and industries can achieve effective disinfection, sanitation, and bleaching, contributing to safer and healthier environments. We encourage readers to delve deeper into the applications of calcium hypochlorite and explore innovative ways to leverage its capabilities to meet their specific needs, thereby maximizing its potential benefits.
[1]https://en.wikipedia.org/wiki/Calcium_hypochlorite
[2] Cui Gang. Maritime safe transportation and management of calcium hypochlorite [J]. World Shipping, 2018, 41(09): 19-22.DOI: 10.16176/j.cnki.21-1284.2018.09.004.
[3]Xu Wenjuan, Chen Yuansheng. Research on the effect of bleaching on the properties of rice paper[J]. Cultural Relics Protection and Archaeological Science, 2007, (02): 20-25. DOI:10.16334/j.cnki.cn31-1652/k.2007.02. 004.
[4]Li Jiangshi. Calcium hypochlorite [J]. Liaoning Chemical Industry, 1991, (04): 60.
[5] Tao Runzhi. Manufacturing and application of calcium hypochlorite [J]. Chemistry World, 1954, (09): 403-404+395. DOI:10.19500/j.cnki.0367-6358.1954.09.007.
 |
 |
 |