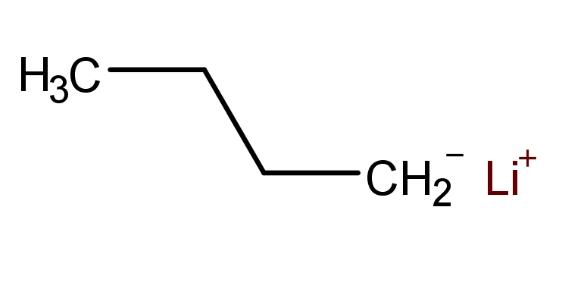
n Butyllithium (n-BuLi) is extensively employed as a polymerization initiator in the production of lithium-based polymers. Industrial applications include the manufacturing processes of thermoplastic elastomers such as SBS, SIS, SEBS, low-cis polybutadiene rubber, solution-polymerized styrene-butadiene rubber, and K-resins. Additionally, n-BuLi finds widespread use in fine chemicals and pharmaceutical industries.
Currently, n Butyllithium is produced under inert gas protection by heating metallic lithium in mineral oil above its melting point to obtain finely dispersed lithium powder with high reactivity. Subsequently, this powder reacts with chloro-n-butane in a solvent system of cyclohexane or n-hexane to produce a solution of n Butyllithium, which is then filtered to isolate the compound.
The physical and chemical properties of n Butyllithium are illustrated in the following diagram:

(1) Alkyl lithium compounds are highly polar due to the carbon-lithium bond, making them strong bases and nucleophilic reagents. n Butyllithium, with a pKa around 50, is commonly used for these properties.
(2) Similar to other alkyl lithium reagents, n Butyllithium's value lies in its ability to generate other functional organolithium compounds through metalation. Metalation reactions rely on the deprotonation ability of alkyl lithium reagents towards weakly acidic protons in aldehydes, ketones, sulfones, nitriles, and other compounds.
(3) The resulting carbanions from deprotonation are valuable intermediates for various chiral transformations and organic reactions. Stable organolithium compounds generated through metalation also serve as nucleophilic reagents in reactions such as reactions with alkyl and alkenyl halides, additions to carbonyl compounds, conjugate additions, and epoxide ring-opening.
(4) Organolithium reagents are also used in the preparation of other organometallic compounds via metal transfer reactions, a useful technique for producing organic copper and titanium compounds, which may exhibit higher selectivity than organolithium compounds.
(5) Industrially, n Butyllithium is crucial as an initiator for anionic polymerization reactions of molecules like butadiene, isoprene, and styrene. These processes are essential for manufacturing various commodities such as rubber tires, plastics, and packaging materials.
(1) While the Occupational Safety and Health Administration has not established permissible exposure limits (PELs), this does not imply n-BuLi's use is without hazards. On the contrary, due to its pyrophoric nature in air and vigorous reaction with water, this chemical poses significant safety risks. This underscores the importance of understanding and adhering to all relevant health and safety aspects of its use.
(2) n Butyllithium is highly destructive to mucous membranes and tissues of the upper respiratory tract, eyes, and skin. Inhalation may lead to bronchospasm, inflammation, and edema, while exposure can cause symptoms such as burning sensation, coughing, wheezing, laryngitis, shortness of breath, headache, nausea, and vomiting.
(1) General advice: Consult a physician. Show this safety data sheet to the attending physician.
(2) If inhaled: Remove to fresh air. If breathing stops, provide artificial respiration. Consult a physician.
(3) If skin contact: Wash thoroughly with soap and plenty of water. Consult a physician.
(4) If in eyes: Rinse cautiously with water for at least 15 minutes. Consult a physician.
(5) If swallowed: Do not induce vomiting. Rinse mouth with water. Consult a physician.
(6) If necessary, provide immediate medical attention and special treatment.
(1) Suitable extinguishing agents: Use water spray, alcohol-resistant foam, dry chemical, or carbon dioxide.
(2) Special protective equipment for firefighters: Wear self-contained breathing apparatus if necessary.
(1) Personal precautions, protective equipment, and emergency procedures: Use personal protective equipment. Avoid dust formation. Avoid inhalation of vapors, mist, or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid inhalation of dust.
(2) Environmental precautions: If safe, prevent further leakage or spillage. Do not allow product to enter drains. Avoid releasing into the environment.
(1) Precautions for safe handling: Avoid contact with skin and eyes. Avoid dust and aerosol formation. Avoid contact - obtain special instructions before use. Provide appropriate exhaust ventilation in places where dust is formed.
(2) Conditions for safe storage, including any incompatibilities: Store in a cool place. Keep container tightly sealed in a dry, well-ventilated place.
Overall, n Butyllithium is considered to be a compound with significant toxicity, necessitating appropriate safety measures during handling and storage. Understanding the potential risks and safety requirements of n Butyllithium is crucial for its use in laboratory environments. By following proper operating procedures and safety precautions, potential hazards can be minimized, ensuring the safety and success of experiments.
[1] http://www.vikit.cn/msds/109-72-8.html
[2] https://en.wikipedia.org/wiki/N-Butyllithium
[3] https://www.fishersci.com/us/en/scientific-products/publications/lab-reporter/2019/issue-2/protecting-n-butyllithium-air-moisture.html
[4] https://enhs.uark.edu/_resources/documents/sops/
[5] Shaoxing Shangyu Hualun Chemical Co., Ltd. A method for preparing n-Butyllithium without mineral oil. June 9, 2023.
 |
 |
 |