n-Butyllithium (n-BuLi), with a molecular formula C4H9Li and a molecular weight of 64.05, is a widely utilized organolithium reagent of significant importance in organic synthesis. Understanding the n butyllithium reaction mechanism is crucial for its applications and modes of action in chemical synthesis.
n-Butyllithium is a potent reagent in organic chemistry, serving as a strong base and nucleophile. It facilitates the formation of reactive organolithium compounds, playing a critical role in various reactions. These organolithium compounds subsequently participate in carbon-carbon bond formations, deprotonations, and other transformations essential for constructing complex molecules.
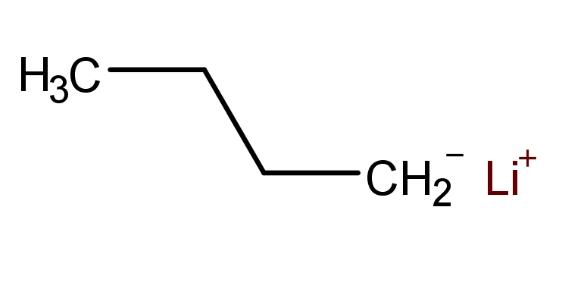
What are the reactions of n-butyllithium? n-Butyllithium functions as a strong base (pKb ≈ -36) and also as a nucleophile and reducing agent, depending on the specific reactants involved. In addition to its role as a strong nucleophile, n-BuLi forms complexes with non-proton Lewis bases such as ethers and tertiary amines, partially decomposing these clusters by binding to the lithium center. Its use as a strong base is termed metalation. Reactions typically occur in tetrahydrofuran (THF) and diethyl ether, which are excellent solvents for the resulting organolithium derivatives.
One of the most useful chemical properties of n-BuLi is its ability to deprotonate (metalate) various types of C-H bonds, especially when conjugate bases are stabilized by electron delocalization or heteroatoms. Examples include alkynes (H-CC-R), methyl sulfides (H-CH2SR), thioacetals (H-CH(SR)2), phosphine methyls (H-CH2PR2), furans, thiophenes, and ferrocene (Fe(H-C5H4)(C5H5)). Additionally, it deprotonates stronger acidic compounds such as alcohols, amines, enolizable carbonyl compounds, and other notably acidic substances, forming lithium alkoxides, amides, enolates, and other salts respectively.
LiC4H9 + RH → C4H10 + RLi
The kinetic basicity of n-BuLi is influenced by solvents or cosolvents. Ligands such as tetrahydrofuran (THF), tetramethylethylenediamine (TMEDA), hexamethylphosphoramide (HMPA), and 1,4-diazabicyclo[2.2.2]octane (DABCO) further polarize the Li-C bonds and accelerate metalation.
An example of using n-BuLi as a base is the addition of amines to methyl carbonate to form methyl carbamate, where n-BuLi is used to deprotonate the amine:
n-BuLi + R2NH + (MeO)2CO → R2NCO2Me + LiOMe + BuH
n-Butyllithium reacts with some organic bromides and iodides to undergo exchange reactions, forming the corresponding organolithium derivatives. This reaction is generally not applicable to organic chlorides and fluorides:
C4H9Li + RX → C4H9X + RLi (X = Br, I)
This lithium-halogen exchange reaction is used to prepare various types of RLi compounds, particularly aryl lithiums and some vinyl lithium reagents. However, the practicality of this method is limited due to the presence of n-BuBr or n-BuI in reaction mixtures, which can react with formed RLi reagents, leading to competitive dehydrohalogenation reactions where n-BuLi acts as a base.

Metal exchange reactions involve the exchange of metals between two organic metal compounds. Many examples of such reactions involve lithium-tin exchanges:
C4H9Li + Me3SnAr → C4H9SnMe3 + LiAr
Organolithium reagents (including n-BuLi) are used to synthesize specific aldehydes and ketones. One synthetic pathway involves the reaction of organolithium reagents with dialkyl amides:
R1Li + R2CONMe2 → LiNMe2 + R2C(O)R1
THF undergoes deprotonation by n-BuLi, especially in the presence of TMEDA, losing a proton adjacent to oxygen. This process consumes n-BuLi to produce butane, initiating ring-opening and forming enolate salts of aldehydes and alkenes. Therefore, reactions of n-BuLi in THF typically occur at low temperatures, such as -78°C, facilitated by dry ice and acetone cooling baths. Higher temperatures (-25°C to -15°C) can also be employed.
Upon heating, n-BuLi, similar to other alkyl lithium reagents with β-hydrogens, undergoes β-hydride elimination, producing 1-butene and lithium hydride (LiH):
C4H9Li → LiH + CH3CH2CH=CH2
An attempt to add functional groups to benzene via deprotonation is typically ineffective with strong bases like alkyl lithium due to kinetic rather than thermodynamic reasons. However, if the benzene ring already contains a group capable of coordinating with the metal of an organometallic, strong bases can induce ring deprotonation, crucial for ortho positions due to coordination with the metal atom.
(1) n-Butyllithium is a pyrophoric reagent and must be handled under strictly dry conditions to prevent ignition upon exposure to air. Typically, solutions of n-BuLi are transferred from their original containers using syringe or cannula techniques into flame-dried reaction vessels under inert gases such as nitrogen or argon.
(2) To prevent exposure of n-BuLi solutions from the original bottle to air, the reagent volumes are replaced with nitrogen or argon. Anhydrous solvents are also used in reactions involving alkyl lithium compounds to minimize degradation of the reagent due to moisture.
(3) Exposure of n-Butyllithium to air or moisture during preparation or storage may lead to decreased concentration, resulting in inaccurate amounts used in reactions. Using partially degraded n-BuLi solutions can lead to lower yields, increased formation of by-products, necessitate more complex and time-consuming purification, and potentially result in synthesis failure.
(4) Packaging of n-Butyllithium should be sufficiently robust to protect the material during transportation while also maintaining the reagent as dry as possible for repeated use.
Understanding the n butyllithium reaction mechanism is crucial for organic synthesis. By comprehending the detailed steps involved, chemists can optimize reaction conditions, predict product outcomes, and develop new synthetic strategies. This knowledge enables finer control over the reactivity of organolithium intermediates, facilitating the more efficient and selective synthesis of desired organic molecules.
[1] https://en.wikipedia.org/wiki/N-Butyllithium#Reactions
[2] https://www.commonorganicchemistry.com/Common_Reagents/n-Butyl_Lithium/n-Butyl_Lithium.htm
[3] https://www.uwindsor.ca/people/jgreen/sites/uwindsor.ca.people.jgreen/files/directed_metallation.pdf
[4] https://macmillan.princeton.edu/
[5] https://www.fishersci.com/us/en/scientific-products/publications/lab-reporter/2019/issue-2/protecting-n-butyllithium-air-moisture.html
 |
 |
 |