Let's delve into the Lewis structure of ethylene (C2H4), a fundamental molecule in organic chemistry. Understanding its Lewis structure provides insights into its bonding pattern, molecular geometry, and properties.

What is the Lewis Structures?
Lewis structures are diagrams that represent the valence electrons in atoms and molecules. They were introduced by Gilbert N. Lewis and are essential tools in understanding chemical bonding. By indicating the arrangement of valence electrons as dots around atoms and the sharing of electrons in bonds, Lewis structures provide a visual depiction of molecular structure and help predict chemical properties.
What is Ethylene (C2H4)?
Ethylene, also known as ethene, is a hydrocarbon with the chemical formula C2H4. It is a colorless, flammable gas with a sweet odor and is widely used in industry, particularly in the production of plastics and as a ripening agent for fruits.
How to draw Lewis structures for Ethylene (C2H4)?
Let's explore the steps to draw the Lewis structure of ethylene:
Step 1: Identify the Central Atom: In ethylene, each carbon atom forms a double bond with another carbon atom. Therefore, both carbon atoms are central.
Step 2: Calculate Total Valence Electrons: Each carbon contributes 4 valence electrons, and each hydrogen contributes 1, giving a total of 4 + 4 + (4 × 1) = 12 valence electrons.
Step 3: Arrange Electrons Around Atoms: Form a double bond between the two carbon atoms, and each carbon atom should have two lone pairs of electrons. Each hydrogen atom should have one lone pair.
Step 4: Fulfill the Octet Rule: Ensure each carbon atom has 8 electrons (2 lone pairs and 2 bonding pairs), and each hydrogen atom has 2 electrons (1 lone pair and 1 bonding pair).
Step 5: Check for Formal Charges: Formal charges may not be necessary as all atoms have achieved the octet rule.

Molecular geometry of Ethylene (C2H4)
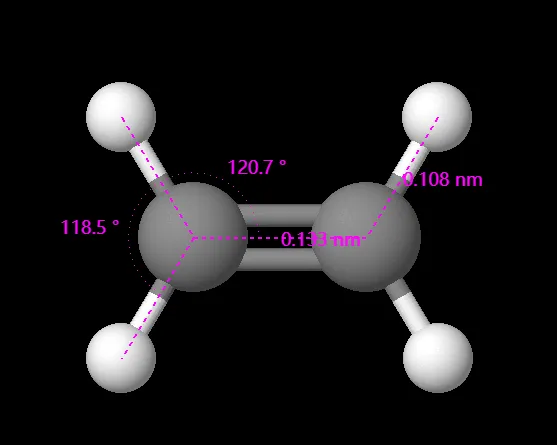
The Lewis structure suggests that ethylene adopts a trigonal planar geometry. In this arrangement, the two carbon atoms lie in the same plane, with each carbon forming three σ bonds, including one double bond between them. The hydrogen atoms occupy positions above and below the plane, resulting in a flat, triangular shape.
Hybridization in Ethylene (C2H4)
In ethylene, each carbon atom undergoes sp2 hybridization. One s orbital and two p orbitals combine to form three sp2 hybrid orbitals, which then overlap with the p orbitals of other atoms to form σ bonds. The remaining p orbital on each carbon atom forms the π bond in the double bond between them. This hybridization ensures the stability and geometry of the ethylene molecule.
Is Ethylene (C2H4) polar or nonpolar?
Ethylene (C2H4) is a nonpolar molecule. Despite containing polar covalent bonds between carbon and hydrogen atoms due to differences in electronegativity, the symmetrical arrangement of atoms in the molecule cancels out any net dipole moment. Therefore, ethylene does not exhibit overall molecular polarity.
What are approximate bond angles and Bond length in Ethylene (C2H4)?
The bond angle in ethylene is approximately 120 degrees. This angle arises from the trigonal planar geometry around each carbon atom, where the hydrogen atoms are positioned at angles of 120 degrees to each other. The bond length of the carbon-carbon double bond is approximately 133 pm, and the carbon-hydrogen bond length is approximately 109 pm.
Note: While VSEPR theory provides a good starting point for predicting molecular geometries and bond angles, real molecules can sometimes deviate from the ideal angles due to factors like lone pair repulsion, bond polarity, and molecular interactions.
Highlight of Ethylene
| ETHYLENE Cas 74-85-1 |
| Molecular formula |
C2H4 |
| Molecular shape |
Trigonal planar |
| Polarity |
nonpolar |
| Hybridization |
sp2 hybridization |
| Bond Angle |
120 degrees |
| Bond length |
C-C: 133 pm C-H: 109 pm |





