
Progesterone, also known as a "female hormone," promotes the maturation of female reproductive organs and the development of secondary sexual characteristics, and maintains normal libido and reproductive function. It can be divided into two main categories (both steroid hormones): estrogens (also called "estrogen hormones") and progestogens. Progestogens are hormones secreted by the luteal cells of the ovaries, mainly progesterone, which is inactivated in the liver to form pregnanediol and is then excreted in the urine after conjugation with glucuronic acid.
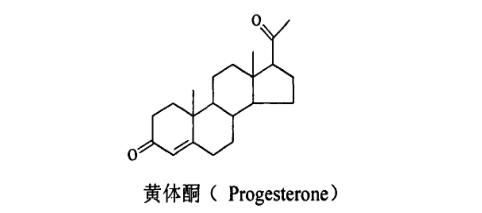
Progesterone is the first natural progestogen discovered, with a chemical structure featuring a C-21 steroid with a 4-3-ketone group. Chemically, progesterone is identical to testosterone's steroid nucleus and Δ4-3-ketone, except that the 17β position of progesterone has an acetyl group, while testosterone has a hydroxyl group. Progesterone was first isolated in pure form in 1934 and later introduced as a drug. Oral Micronized Progesterone (OMP) was released in 1980, allowing for oral administration of progesterone.
Micronized Progesterone is a prescription medication used to prevent endometrial hyperplasia and treat symptoms of secondary amenorrhea (absence of menstruation). Micronized Progesterone is available under brand names such as Prometrium. Micronized Progesterone is a bioidentical hormone with the same molecular structure as endogenous progesterone produced by the ovaries. In contrast, synthetic progestogens have different chemical structures compared to progesterone.
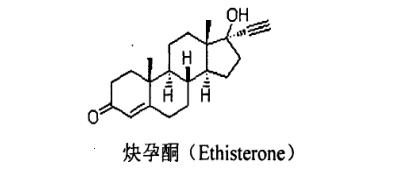
In research on oral progestogens, the first effective oral medication was not a progesterone derivative but rather a derivative of testosterone—Norethindrone. After introducing an acetylene group at the 17α position, its androgenic activity was reduced, revealing progestogenic activity and effectiveness when taken orally. Although Norethindrone is 15 times more active orally than progesterone, it still retains about 1/10th of testosterone's androgenic activity, which can lead to side effects in women.
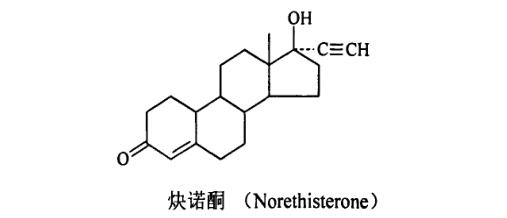
In 1954, Djerassi and colleagues further modified Norethindrone's structure by removing the methyl group at position 19, resulting in Norethindrone, which showed a 5-fold increase in oral progestogenic activity compared to Norethindrone and only 1/20th of testosterone's androgenic activity. As a result, Norethindrone, introduced in 1956, has been widely used. By 1963, China successfully tested Norethindrone, and it has been used as a contraceptive in clinical trials. Currently, pharmacopeias in China, the United States, the United Kingdom, and Japan list Norethindrone. Norethindrone is a white or off-white crystalline powder, odorless, slightly bitter, soluble in ethanol, slightly soluble in acetone, and insoluble in water. It has a melting point of 202-208°C.
Progestogens are compounds with progestogenic activity, inducing the secretory endometrium to support pregnancy. Post-ovulation, endogenous progestogen levels rise to prepare the endometrium for implantation and support it during pregnancy (when circulating progestogens reach typical high levels). However, recently the term "progestogen" is often used specifically to describe synthetic progestogens such as MPA, Norethindrone, and Levonorgestrel, excluding natural progesterone. The term "progestogen" aligns with the naming conventions for other hormone groups like androgens and estrogens, which are defined by their androgenic and estrogenic activities, respectively. To avoid confusion, the North American Menopause Society recommends using the term "progestogen" for synthetic progestogens and "progesterone" exclusively for natural progesterone.
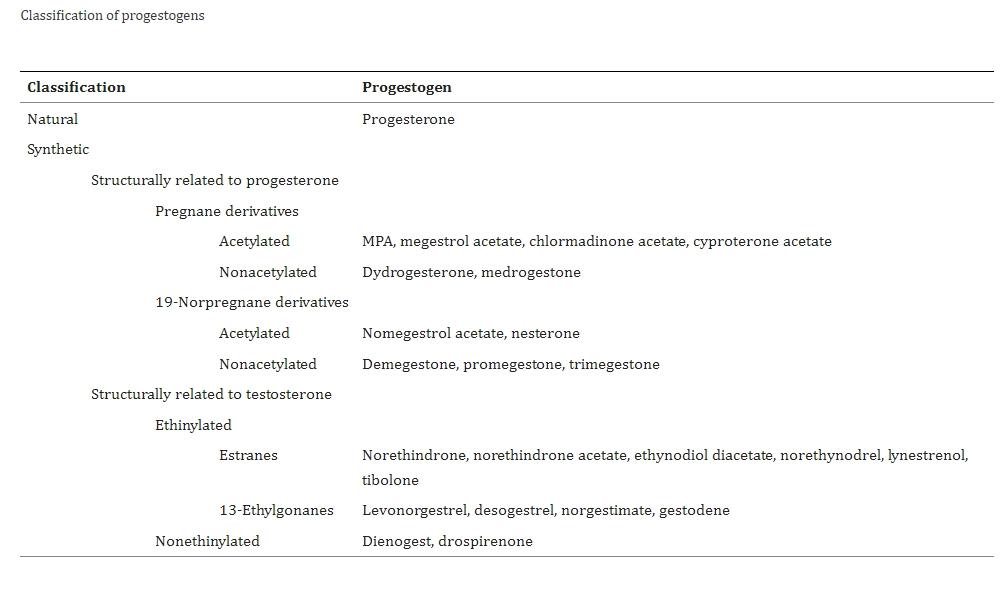
Progestogens can be classified into two types: natural progestogen and synthetic progestogens. As mentioned, natural progestogen is only progesterone. In contrast, multiple synthetic progestogens are available for treatment, differing greatly in chemical structure. These are categorized into: 1) those structurally related to progesterone; and 2) those structurally related to testosterone.
Norethindrone is a synthetic progestogen, biochemically similar to testosterone—more so than other progestogens. Compared to Micronized Progesterone, Norethindrone has a slightly higher risk of breast cancer but effectively controls troublesome bleeding and is conveniently used in combination pills.
The use of non-estrogenic progestogens is associated with high risks of endometrial hyperplasia and adenocarcinoma (relative risk of 2.1 to 5.7). It has long been recognized that adding progestogens for at least 10 to 14 days each month is necessary to prevent these effects. However, synthetic progestogens like Norethindrone and MPA have been linked with metabolic and vascular side effects (e.g., inhibiting estrogen's vasodilatory effects) in experimental and clinical studies. Comparisons have concluded that using natural progestogens can minimize or eliminate side effects associated with synthetic progestogens, as natural progestogens are identical to steroids produced by the corpus luteum. Oral Micronized Progesterone avoids the inconvenience of injectable, rectal, or vaginal preparations. Its bioavailability is similar to other natural steroids, with variability in area under the curve being comparable to synthetic progesterone. A significant dose-range effect has been demonstrated, with long-term protective effects on the endometrium. Since 1980, Micronized Progesterone has been widely used in Europe, with doses ranging from 300 mg/day (taken at bedtime) for women desiring monthly bleeding to 200 mg every 14 days or 100 mg every 25 days for women wishing to remain amenorrheic.

There are many different types of progestogens (synthetic progesterones) available for women. Research suggests that Micronized Progesterone (known as Utrogestan in the UK) is considered the best type of progestogen. This progesterone has a molecular structure identical to natural progesterone in the body, usually resulting in fewer side effects compared to other progestogen forms. Compared to synthetic progestogens like Provera (MPA), Micronized Progesterone produces nearly no side effects. Additionally, postmenopausal women taking this bioidentical progestogen can effectively reduce the risk of osteoporosis.
Alina Warenik-Szymankiewicz and colleagues assessed the effects of vaginal Micronized Progesterone and compared it to oral Norethindrone. They measured lipid profiles, carbohydrate parameters, and coagulation factors. The conclusion was that vaginal Micronized Progesterone is well-tolerated and effective in menopause. Comparisons of Micronized Progesterone and Norethindrone in hormone replacement therapy showed no statistically significant differences in their effects on lipid and carbohydrate metabolism and coagulation factors.
When choosing between Norethindrone and Micronized Progesterone, understanding their respective advantages and limitations is crucial. Each medication has specific indications and side effects. Therefore, it is recommended to consult with a doctor before deciding which medication to use. A healthcare provider can offer the most appropriate advice and treatment plan based on individual health conditions and needs to ensure the selection of the most effective and safe medication. Avoid self-prescribing and make an informed decision through professional medical advice.
[1]Warenik-Szymankiewicz A, Hada? K. Therapeutic effectiveness–comparison of progesterone and norethisterone in hormonal replacement therapy in women with hormonal disturbances in climacteric period[J]. Menopause Review/Przegl?d Menopauzalny, 2004, 3(6): 27-32.
[2]https://en.wikipedia.org/wiki/Progesterone_(medication)
[3]https://www.ncbi.nlm.nih.gov/books/NBK563211/
[4]https://www.rxlist.com/progesterone_micronized/generic-drug.htm
[5]https://pubmed.ncbi.nlm.nih.gov/10090424/
[6]https://www.clinicaltherapeutics.com/article/S0149-2918(00)88267-3/abstract
[7]https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4960754/
[8]Stanczyk F Z, Hapgood J P, Winer S, et al. Progestogens used in postmenopausal hormone therapy: differences in their pharmacological properties, intracellular actions, and clinical effects[J]. Endocrine reviews, 2013, 34(2): 171-208.
[9]Shi Cheng. Study on the synthesis process of norethindrone[D]. Zhejiang: Zhejiang University, 2010. |
 |
 |