Oleylamine, with the chemical formula CH3(CH2)7CH═CH(CH2)8NH2, is a linear unsaturated primary amine that remains liquid at 25°C due to its double bond. Economical commercial oleylamine reagents are widely used in the synthesis of metals, semiconductors, metal halide perovskites, and nanomaterials. Oleylamine serves as a solvent, electronic surface passivation ligand, and long-chain surfactant, stabilizing colloids across a broad temperature range. It is often referred to as a key coordinating solvent in nanomaterial synthesis. The CAS nomenclature for oleylamine is cis-9-octadecen-1-amine (Oleylamine cas number 112-90-3).
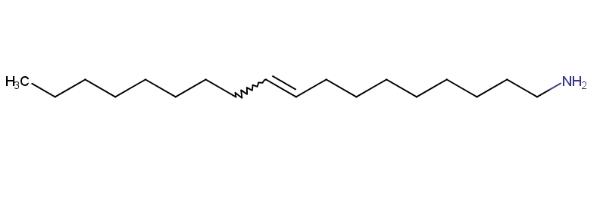
Oleylamine has the molecular formula C18H37N and a molecular weight of 267.49 g/mol. Its chemical structure includes a long alkyl chain (18 carbon atoms) and an amino (-NH2) group. The alkyl chain of oleylamine is linear and unbranched, while the presence of a double bond makes it an unsaturated fatty amine. The position of the double bond in the middle of the chain imparts certain double-bond characteristics to oleylamine. The length of the chain and the double bond contribute to oleylamine's fluidity and flexibility. The chemical structure of oleylamine is as follows:
Oleylamine Solubility in Other Solvents: Soluble in alcohols, chloroform, hydrocarbons
Oleylamine reacts with carboxylic acids to form carboxylates through an exothermic reaction. The carboxylates can further condense into amides by losing a water molecule. In the presence of acetic acid, oleylamine forms insoluble complexes with DNA, with particle radii ranging from 60 to 65 nm.
The pKa of oleylamine is predicted to be 10.67±0.10. Oleylamine is classified as an amine, which is a type of chemical base that reacts with acids to form salts and water, typically releasing heat. The heat released per mole of amine during neutralization is not strongly related to its basicity. Oleylamine may be incompatible with isocyanates, organic halides, peroxides, acidic phenols, epoxides, anhydrides, and acid chlorides. Furthermore, oleylamine can generate flammable hydrogen gas when reacting with strong reducing agents such as hydrides.
Oleylamine, with its excellent chemical properties and broad application potential, has become a crucial component in many industrial and research fields. Its use in materials science, chemical synthesis, and as a surfactant showcases its unique advantages. For a deeper understanding of oleylamine's supply and procurement options, it is recommended to visit the Guidechem website. On Guidechem, you can find various oleylamine suppliers and obtain the latest market information to make informed purchasing decisions.
 |
 |
 |