
Paraffin wax, or alkanes, is a straight-chain or branched-chain saturated organic compound with the general formula CnH2n+2. The term paraffin wax refers to a mixture of various hydrocarbons, particularly alkanes and cycloalkanes, that are solid at room temperature. Paraffin wax is abundant in nature but can also be artificially synthesized and forms as a byproduct of processing certain natural substances. Paraffin wax is solid at room temperature and can be obtained from lubricating oil fractions with different average boiling points, distillation residues from vacuum distillation of hydrocarbon crude oil, and so-called tank wax and pipeline wax separated during the storage and transportation of such crude oil. Crude oil and its products contain significant amounts of individual paraffin wax. Even lower boiling petroleum fractions contain varying amounts of other hydrocarbons, such as cycloalkanes and aromatic compounds, depending on the source of the crude oil.
Paraffin wax is a solid crystalline mixture of straight-chain (normal) hydrocarbons with molecular chain lengths ranging from C20 to C30 or even higher, represented by CH3(CH2)nCH3, where n ≥ 18. The molecular formula of paraffin wax is expressed by the general formula CnH2n+2, where n is a large number, typically between 20 and 40. Paraffin wax consists of a mixture of alkanes (straight-chain hydrocarbons) of different chain lengths. These alkanes are characterized by carbon atoms in the chain connected by single covalent C-C bonds. This straight-chain, unbranched structure imparts the rigidity to paraffin wax.
Paraffin wax is mainly classified into two types: macrocrystalline wax and microcrystalline wax. Macrocrystalline wax is a mixture of saturated normal C18–C30 hydrocarbons and small amounts of isoalkanes and cycloalkanes. The molecular weight of the components ranges from 250 to 400, paraffin wax melting point between 40–60°C. The crystals are either plate-like or needle-like. Microcrystalline wax, in addition to containing normal alkanes, also contains a significant amount of isoalkanes and cycloalkanes with long alkyl side chains. Isoalkanes form microcrystals, and the main components of these waxes are C40–C55 compounds. Paraffin wax with a macrocrystalline structure can be classified based on its melting point or refining degree.
Paraffin wax is mostly a white, odorless, tasteless, waxy solid with a typical melting point of about 46 to 68°C (115 to 154°F) and density of paraffin wax approximately 900 kg/m3. It is characterized by being solid at room temperature (25°C, 77°F) and having low viscosity when melted (viscosity is 35–45 SUS at 99°C, 210°F). It is insoluble in water but soluble in ether, benzene, and some esters. Paraffin wax is unaffected by most common chemical reagents, but is highly flammable. Its heat of combustion is 42 MJ/kg.
Paraffin wax is an excellent thermal storage material. Heat capacity of paraffin wax is 2.14-2.9 J/g·K (Joules/gram/Kelvin) and a heat of fusion of 200-220 J/g. Paraffin wax's phase change cooling, combined with expandable heat exchangers, was used to cool the electronics of lunar rovers in manned moon missions in the early 1970s. Wax significantly expands when melted, making it useful in industrial, domestic, and especially automotive applications, such as thermostats.
Paraffin wax can serve as an excellent phase change material due to its high latent heat of fusion, negligible supercooling, low vapor pressure in the molten state, chemical inertness, and stability. The number of carbon atoms in paraffin wax chains ranges from 18 to 50 (C18–C50) with melting temperatures between 30°C and 90°C. The specific heat capacity of latent heat paraffin wax is approximately 2.1 kJ/kg·°C. Its heat of fusion ranges from 180 to 230 J/g, which is relatively high for organic materials. This combination results in excellent energy storage density.
The melting point of paraffin wax ranges from about 48 to 66°C (120° to 150° F). The length of the paraffin wax chain varies, determining the melting temperature and thus the phase change. Phase change can be promoted by altering pressure and temperature conditions. As a phase change polymer, the mechanical properties of paraffin wax are related to its thermal properties. As mentioned, paraffin wax with different chain lengths has a range of melting temperatures and undergoes solid-liquid phase changes, with interphase volume expansion varying significantly, ranging from 5-40% (Carlen, 2001).
Paraffin wax is abundant in nature but can also be artificially synthesized and forms as a byproduct of processing certain natural substances. What is paraffin wax made from? Paraffin wax was first commercially produced in 1867 by cooling petroleum to yield paraffin wax precipitation. Today, paraffin wax is obtained by dewaxing light lubricating oil feedstock from petroleum.
As early as 1867, shortly after the successful drilling of the first oil wells, paraffin wax achieved commercial production. Because paraffin wax is easily precipitated from petroleum during cooling, early technological innovations focused on improving the separation and purification efficiency of paraffin wax. Traditional paraffin wax purification processes involve a series of complex operations, including chemical treatment, adsorption decolorization, and physical methods such as distillation and recrystallization. The variation in paraffin wax content across different crude oils introduces diversity into paraffin wax production processes.
Synthetic paraffin wax is a byproduct of the conversion of coal gas into hydrocarbons through Fischer-Tropsch synthesis. Since they exist as a mixture of straight-chain or branched saturated alkanes, no further polymerization is required. Post World War II, synthetic paraffin wax entered the commercial market as a product of the Fischer reaction, which converts coal gas into hydrocarbons. Compared to natural paraffin wax, synthetic paraffin wax exhibits higher purity and superior properties, such as increased whiteness and hardness. These characteristics make it suitable for a wide range of applications, including as a substitute for some plant waxes, and for modifying petroleum wax and certain plastics such as polyethylene. The advent of synthetic paraffin wax further enriched the variety of paraffin wax products, meeting the specific needs of different industrial sectors.
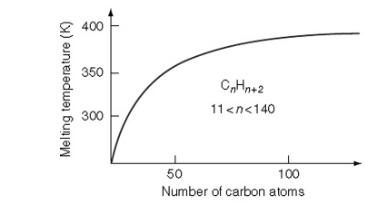
Paraffin wax consists of large molecules of hydrogen and carbon. Due to the small electronegativity difference between hydrogen and carbon, paraffin wax exhibits a nonpolar covalent character. Its main components are alkanes, which interact through dispersion forces, forming nonpolar molecules. As the carbon chain length increases, these dispersion forces strengthen, resulting in higher melting and boiling points for paraffin wax.
[1]https://en.wikipedia.org/wiki/Paraffin_wax
[2]https://www.britannica.com/science/paraffin-wax
[3]https://www.sciencedirect.com/topics/chemical-engineering/paraffin-wax
[4]https://www.sciencedirect.com/science/article/abs/pii/S1359836813000139
[5]https://www.sciencedirect.com/science/article/pii/B9780128035818005221
 |
 |
 |