
Paraquat dichloride, also known as methyl violet, has the molecular formula C12H14Cl2N2 and is a fast-acting, non-selective herbicide. Paraquat dichloride toxicity is highly to mammals; it is a highly toxic contact herbicide that rapidly destroys chloroplast membranes, quickly halting photosynthesis and chlorophyll synthesis in green plants. It is also known commercially as Gramoxone or Paraquat. Soluble in water, the paraquat available on the market is usually a 20% solution, odorless, dark green, corrosive, and stable in acidic conditions but decomposes in alkaline environments. Its chemical structure is 1,1’-dimethyl-4,4’-bipyridinium dichloride. Paraquat dichloride loses its activity upon contact with soil and does not pollute the environment, which is why it is widely used in agriculture. However, paraquat dichloride is highly toxic to humans and animals, with a lethal dose for adults being around 5-15 ml of a 20% solution or 40 mg/kg. Since the first case of paraquat dichloride poisoning was reported in the UK in the 1960s, thousands have died from this pesticide, primarily due to oral ingestion. Paraquat dichloride can enter the body through three routes: oral ingestion, skin absorption, and inhalation.
Amoebic cells actively uptake and accumulate paraquat. Paraquat primarily accumulates in type I and II alveolar cells, and high concentrations of paraquat accumulate in lung and kidney cells, disrupting cellular redox reactions and generating harmful oxygen free radicals. This process damages cellular defense mechanisms, leading to lung damage and renal tubular necrosis.
The main target organs for paraquat poisoning are the lungs and kidneys. The structure of paraquat is similar to that of natural polyamines absorbed by alveolar cells, leading to its concentration in alveolar type I and II cells. Paraquat is also actively secreted by the kidneys, resulting in high concentrations in proximal tubular epithelial cells. After accumulation in the alveoli and renal units, paraquat induces oxidative stress and produces toxic reactive oxygen species. This oxidative stress overwhelms cellular defenses and causes lung damage (alveolitis and fibrosis). At moderate doses, initial lung damage progresses to pulmonary fibrosis due to the rapid proliferation and differentiation of fibroblasts, leading to the loss of lung structure. Paraquat causes vacuolation of proximal renal tubular cells, leading to tubular necrosis. Liver cell damage results from mitochondrial damage and endoplasmic reticulum degranulation.
Paraquat is toxic to humans when ingested orally (Class II), and has moderate toxicity when absorbed through the skin (Class III). Ingesting pure paraquat is extremely toxic to mammals, including humans, causing severe inflammation and potentially severe lung damage (e.g., irreversible pulmonary fibrosis, also known as "paraquat lung"), acute respiratory distress syndrome (ARDS), and death. The mortality rate is estimated between 60-90%.
Inhalation of paraquat is highly toxic, with acute inhalation toxicity rated as Class I, the highest danger level. Although the EPA stipulates that particles used in agriculture (400-800 μm) are not within the inhalable range, paraquat still causes moderate to severe irritation to the eyes and skin. Although diluted paraquat has lower toxicity when sprayed, the main risk of accidental poisoning occurs during the mixing and loading of paraquat.
Paraquat is a corrosive herbicide and can cause gastrointestinal corrosion upon ingestion. Oral and tongue mucosal lesions are known as "paraquat tongue," appearing within the first few days and potentially leading to ulceration and bleeding. These lesions are not prognostically significant, as even small amounts of paraquat can cause them if ingested. Esophageal ulcers cause pain and difficulty swallowing, which can develop into perforation, mediastinitis, and mediastinal emphysema. Other routes of paraquat exposure include skin/eye contact and inhalation. Intact skin is an effective barrier against absorption, with skin contact being life-threatening primarily in cases of existing skin lesions or prolonged exposure (e.g., wearing clothing soaked in spray). Local effects include skin irritation, blistering, and full-thickness burns. Inhalation of paraquat mist (where the recommended spray droplets are too large to inhale) may cause respiratory exposure. Inhalation of sprayed paraquat solutions usually causes local irritation, but rarely leads to significant systemic absorption. To prevent accidental ingestion or reduce paraquat absorption, many manufacturers add dyes, deodorizers, and emetics to concentrated solutions.
Clinical features of paraquat poisoning depend on the dose, classified into mild, moderate, and severe poisoning. The following table shows the classification of paraquat poisoning based on severity.
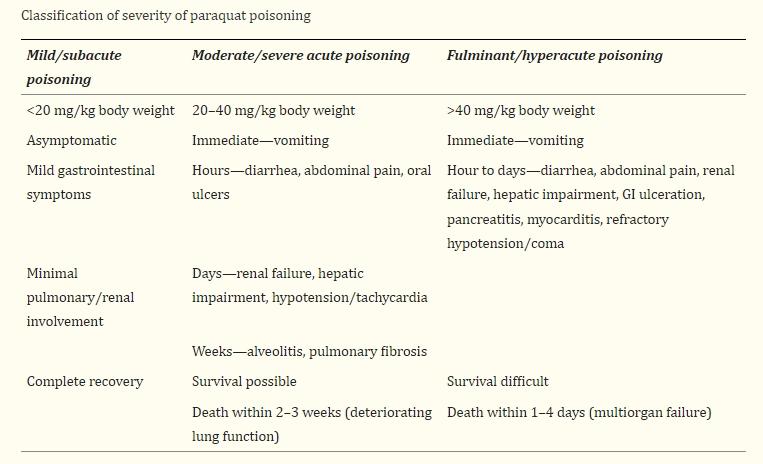
After ingesting a large amount of paraquat, an individual is likely to experience immediate pain and swelling of the mouth and throat. The next signs of illness are gastrointestinal symptoms such as nausea, vomiting, abdominal pain, and diarrhea (which may become bloody). Severe gastrointestinal symptoms may lead to dehydration (fluid deficiency), electrolyte imbalance (deficiency of sodium and potassium), and hypotension (low blood pressure) (CDC).
Ingesting small to moderate amounts of paraquat may lead to the following adverse health effects within days to weeks:
Generally, ingesting large amounts of paraquat leads to the following signs/symptoms within hours to days:
Long-term Health Effects
If a person survives paraquat dichloride poisoning, they are likely to suffer from long-term lung damage (scarring). Other long-term effects may include kidney failure, heart failure, and esophageal strictures (scarring of the swallowing tube making it difficult to swallow). Individuals who ingest large amounts of paraquat are unlikely to survive (CDC).
There is no specific antidote for paraquat dichloride poisoning. Treatment principles include:
reducing toxin absorption;
removing free toxins as quickly as possible while protecting vital organs;
preventing lung damage and fibrosis.
The standard treatment for paraquat dichloride poisoning involves gastric lavage to remove as much of the poison as possible. Bleaching agents or activated charcoal may also improve treatment outcomes, depending on the timing of administration. Blood dialysis, blood filtration, blood perfusion, or antioxidant therapy may be recommended. Some suggest using immunosuppressive therapy to reduce inflammation, but there is only low-certainty evidence supporting the combined use of glucocorticoids and cyclophosphamide to lower mortality. It is currently unclear whether adding glucocorticoids and cyclophosphamide to standard treatment increases the risk of adverse side effects such as infections. Oxygen should not be administered unless SpO2 levels are below 92%, as high concentrations of oxygen can exacerbate toxic effects. Patients may die up to 30 days after ingestion.
The risk of paraquat dichloride poisoning should not be underestimated, and its severity requires high vigilance. Prompt and correct emergency and medical interventions are crucial in poisoning incidents. Understanding and following the guidelines in the paraquat dichloride Safety Data Sheet (SDS) and Material Safety Data Sheet (MSDS) is an effective means of preventing and managing poisoning. Only with comprehensive knowledge of these safety information and rapid response can the harm of paraquat dichloride poisoning be minimized, ensuring the safety and health of individuals.
[1]Li Bing. Expression of cytokines in the kidneys of rats with acute paraquat poisoning and the effect of ulinastatin on them[D]. Hebei: Hebei Medical University, 2018. DOI:10.7666/d. D01513875.
[2]https://www.sciencedirect.com/science/article/abs/pii/0041008X87902833
[3]Sukumar C A, Shanbhag V, Shastry A B. Paraquat: The poison potion[J]. Indian journal of critical care medicine: peer-reviewed, official publication of Indian Society of Critical Care Medicine, 2019, 23(Suppl 4): S263.
[4]https://emergency.cdc.gov/agent/paraquat/basics/facts.asp
[5]https://baike.baidu.com/item/%E7%99%BE%E8%8D%89%E6%9E%AF
[6]https://baike.baidu.com/item/%E7%99%BE%E8%8D%89%E6%9E%AF%E4%B8%AD%E6%AF%92%E6%B2%BB%E7%96%97
[7]https://en.wikipedia.org/wiki/Paraquat
[8]https://pubchem.ncbi.nlm.nih.gov/compound/Paraquat
[9]https://www.webmd.com/a-to-z-guides/what-to-know-about-paraquat-poisoning
 |
 |
 |