What is Phenylmagnesium Chloride?
Phenylmagnesium chloride is an intermediate for the preparation of triphenylphosphine, which is a fundamental raw material for rhodium-phosphine complex catalysts, widely used in petrochemicals. Triphenylphosphine is also applied in pharmaceuticals, organic synthesis, analysis, and as a Wittig reagent. Additionally, it serves as a brightener in dye processes, a stabilizer in polymerization, an antioxidant in film development, and can be used as an analytical reagent.
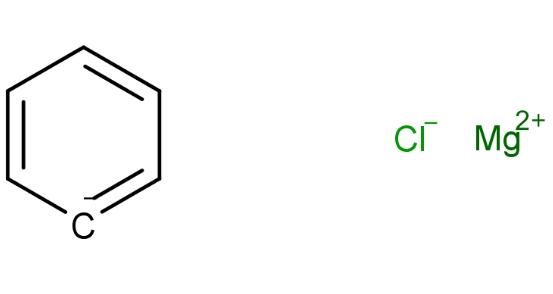
Molecular Formula and Structure Analysis of Phenylmagnesium Chloride
Phenylmagnesium chloride formula is C6H5ClMg, with a molecular weight of 136.86. It consists of 52.66% carbon, 3.68% hydrogen, 25.90% chlorine, and 17.76% magnesium. The molecular structure of Phenylmagnesium chloride includes a central magnesium (Mg) atom bonded to a chlorine (Cl) atom and a phenyl group (C6H5). The Mg-Cl bond exhibits significant ionic character, while the Mg-C bond shows a combination of ionic and covalent characteristics due to the electronegativity difference between magnesium and carbon. The molecule has a trigonal planar geometry around the magnesium center. The structure of Phenylmagnesium chloride is as follows:
Properties
- Form: Liquid
- Color: The solution is light yellow-brown to dark brown
- Phenylmagnesium chloride density: 0.98 g/mL at 20°C
- Boiling Point: 78.8°C at 760 mmHg
- Melting Point: 89-90°C
- Flash Point: 1°F
- Water Solubility: Reacts violently with water
- Vapor Pressure: 101 mmHg at 25°C
- Stability: Unstable; reacts readily with water vapor and carbon dioxide when exposed to air
- Storage Conditions: Store at 2-8°C, protected from light, and away from strong oxidizers
Safety and Hazards of Phenylmagnesium Chloride
Phenylmagnesium chloride reacts with water to release toxic and flammable gases, generating heat; this reaction is quite vigorous. Phenylmagnesium chloride is highly flammable and strongly irritates the skin and eyes. Contact may result in severe eye damage and liver injury. It may also be absorbed through the skin.
Hazard Identification
Classification of Substance or Mixture:
Flammable liquids, Category 2; Skin corrosion, Category 1B; Serious eye damage, Category 1
GHS Label Elements:Symbols:

Signal Word:
Danger
Hazard Statements:
- H225 (75.47%): Highly flammable liquid and vapor [Danger Flammable liquids]
- H314 (100%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
- H318 (56.6%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
First Aid Measures
- General Advice: Consult a physician. Show the safety data sheet to the attending physician.
- If Inhaled: Move the person to fresh air. If breathing stops, perform artificial respiration. Consult a physician.
- If Skin Contact Occurs: Wash with soap and plenty of water. Consult a physician.
- If Eye Contact Occurs: Rinse with plenty of water for at least 15 minutes and consult a physician.
- If Swallowed: Do not induce vomiting if the person is unconscious. Rinse mouth with water. Consult a physician.
Handling and Storage
- Safe Handling Precautions: Avoid contact with skin and eyes. Avoid forming dust and aerosols. Obtain special instructions before use. Provide adequate ventilation in areas where dust may be formed.
- Safe Storage Conditions, Including Incompatibilities: Store in a cool place. Keep the container tightly closed in a dry, well-ventilated area.
Reactivity and Stability
- Chemical Stability: Stable under recommended storage conditions.
- Possibility of Hazardous Reactions: Reacts with water, steam, or acids to produce toxic and flammable vapors and heat.
References
[1]Zhejiang Wansheng Co., Ltd. A preparation process of phenylmagnesium chloride: CN202310987013.1[P]. 2023-11-10.
[2]http://www.vikit.cn/msds/100-59-4.html
[3]https://pubchem.ncbi.nlm.nih.gov/compound/7513
[4]https://merckindex.rsc.org/monographs/m8679
[5]https://baike.baidu.com/item/%E8%8B%AF%E5%9F%BA%E6%B0%AF%E5%8C%96%E9%95%81




