
Phthalonitrile, also known as phthalocyanine or 1,2-dicyanobenzene, is a synthetic aromatic macrocyclic compound. Although its structure is similar to that of natural porphyrins, it does not exist in nature. Due to its structure and electronic properties, phthalonitrile has found wide applications in various fields. Utilizing its absorbance in the red region of the UV-visible spectrum (typically between 620 and 700 nm), phthalonitrile has been successfully used or tested as a photosensitizer (PS) in various medical applications, including photodynamic therapy (PDT) for cancer treatment, photodynamic inactivation (PDI) of microorganisms, and as a diagnostic agent. Phthalonitrile is also being studied as a potential drug for treating amyloid-related neurodegenerative diseases. It is frequently reported for use in chemical sensors, microbial fuel cells, DNA interactions, catalysis, and solar cells. Functionalization of the phthalonitrile macrocycle can adjust its properties and design compounds with new potentials. For example, incorporating sulfonamide (SA) groups into the phthalocyanine periphery to enhance the efficacy and selectivity of bacterial targeting is a modification of this type.
Phthalonitrile is an important organic intermediate used widely for synthesizing high-heat-resistant polyamides and polyesters, a series of phthalocyanine pigments, novel dimethylphenyl diisocyanate plastics, and desulfurization catalysts. For instance, reacting phthalonitrile with copper or copper chloride can produce phthalocyanine pigment precursors, which can then be used to synthesize a range of phthalocyanine pigments. Phthalocyanine pigments have developed rapidly in developed countries and are widely used.
There are several options for synthesizing phthalonitrile:
To address issues such as low yield and poor product color in ortho-xylene ammonia oxidation, Cong Linquan and colleagues selected phthalic anhydride as the raw material, with self-made solid acid as the catalyst, and continuous atmospheric pressure fixed-bed dehydration reaction. Under optimal conditions, the yield of phthalonitrile reached 96.2%. The experimental process is as follows:
Phthalonitrile Synthesis Reaction
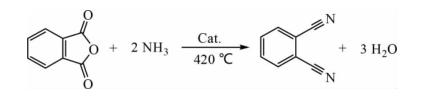
Using self-made solid acid catalyst and phthalic anhydride as raw material to produce phthalonitrile at a reaction temperature of 420 °C, with phthalic anhydride conversion reaching 100% and molar yield of phthalonitrile reaching 96.2%.
The experimental setup, as shown in the figure, involves ammonia gas after vacuum treatment entering the material mixer through a rotor flowmeter, mixing with phthalic anhydride from a metering pump, and then entering a fixed-bed reactor. The fixed bed maintains a constant temperature zone with a catalyst, and the mixed gas reacts in the catalyst bed, with the resulting product collected in a receiver. The exhaust gas is absorbed by acid solution before being discharged. The reaction tube is a stainless steel pipe with an outer diameter of 25 mm and an inner diameter of 22 mm, heated by an electric furnace.
To address the formation of phthalimide in ortho-xylene ammonia oxidation, Ma Yulong and colleagues selected a highly selective ODT-88 catalyst. This catalyst was used to study reaction conditions such as the ratio of air to ammonia and temperature. Under optimized conditions, the molar yield of phthalonitrile reached 94.4%.
Mix a specified amount of V2O5, reduce with oxalic acid until a deep blue oxalic acid vanadium solution is obtained, then add appropriate amounts of Cr2O3 and P2O5. After making a solution, add SiO2 and allow the solution to fully impregnate SiO2, then dry at 100 °C and activate at 650 °C.
The experimental setup, as shown in the figure, involves air and vacuum-treated ammonia gas measured separately entering the material mixer, mixing with ortho-xylene from a metering pump, and then entering a fixed-bed reactor. The lower part of the fixed-bed reactor contains the catalyst, and the mixed gas reacts in the catalyst bed, with the resulting product collected in a collector. The remaining exhaust gas is treated with alkaline and water washes before being discharged (not shown in the figure). The fixed-bed reactor is made of a Φ33 mm quartz tube, heated by a tubular electric furnace with a slightly larger inner diameter than the reactor's outer diameter.
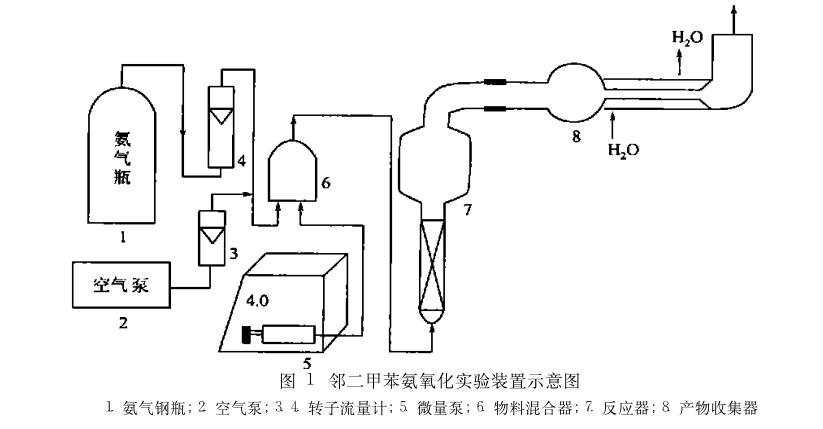
The product is analyzed by gas chromatography, with qualitative and quantitative correction based on known substances. The components and quantities of the collected material in the collector vary depending on the catalyst and reaction conditions used, with main by-products including ortho-toluene nitrile, phthalimide, and starting material ortho-xylene.
 |
 |