Calcium hypochlorite is a white powdered solid with a strong irritating chlorine odor, primarily produced using chlorine gas and calcium hydroxide (lime) as raw materials. It undergoes strict control of reaction conditions and separation purification in electrolytic cells or oxygen reactors. It is a calcium salt and inorganic calcium salt with bleaching properties containing hypochlorite. This comprehensive guide provides readers with a thorough understanding of the calcium hypochlorite formula, composition, properties, applications, and safety precautions, ensuring its safe and effective use across various industries.
(1) What is the molecular formula of calcium hypochlorite?
Calcium hypochlorite, an important compound in various industries, consists primarily of calcium, chlorine, and oxygen. The molecular formula of calcium hypochlorite is Ca(ClO)2. Its molecular structure consists of calcium cations (Ca2 +) combined with hypochlorite anions (ClO?). The formulation of calcium hypochlorite typically involves incorporating these ions into solid granules or powder forms soluble in water. In aqueous solution, calcium hypochlorite undergoes decomposition, releasing hypochlorous acid (HOCl) and calcium hydroxide (Ca(OH)2), both of which contribute to its disinfection and bleaching properties.
(2) Comparison with other chlorinated compounds
Compared to other chlorinated compounds, calcium hypochlorite stands out for its stability and versatility. Unlike sodium hypochlorite commonly found in household bleach and liquid disinfectants, calcium hypochlorite offers greater stability and a longer shelf life, making it more suitable for long-term storage and transportation. With higher chlorine content compared to sodium hypochlorite, calcium hypochlorite exhibits better disinfection efficacy per unit compound.
(1) Detailed breakdown of elements in the formula
The structure of the Ca(ClO)2 molecule is illustrated below. It consists of one calcium ion and two hypochlorite ions.
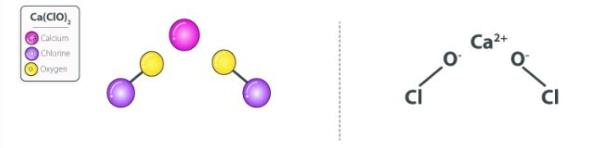
Each hypochlorite ion carries a -1 charge while the calcium ion carries a +2 charge. Thus, one calcium ion can form ionically bonded with two hypochlorite ions.
(2) Preparation
Calcium hypochlorite is industrially produced by the reaction of chlorine gas with calcium hydroxide. The chemical equation for this reaction is as follows:
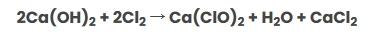
In general, bleaching powder is a mixture of calcium hypochlorite, dihydrate (Ca(ClO2).2Ca(OH)2), and calcium chloride dihydrate (CaCl2.2Ca(OH)2). Due to its two chlorine ions, this compound has higher chlorine availability compared to sodium hypochlorite (NaOCl).
Calcium hypochlorite undergoes several chemical reactions crucial for its disinfection and bleaching action. When dissolved in water, calcium hypochlorite dissociates into calcium ions (Ca2 +) and hypochlorite ions (ClO-). Hypochlorite ions readily react with water molecules to produce hypochlorous acid (HOCl) and hydroxide ions (OH -). Hypochlorous acid is a potent oxidizing agent, effectively neutralizing various pathogens by disrupting their cellular structures and metabolic processes. Additionally, hypochlorous acid plays a crucial role in the bleaching process, oxidizing pigments and chromophores in stains and colored compounds, rendering them colorless and improving the appearance of fabrics and surfaces.
Chemical Properties

Physical Properties
(1) Calcium hypochlorite is a white/gray solid at room temperature.
(2) Solubility in water is 21g/100ml, undergoing reaction upon dissolution.
(3) Its solubility in hard water is relatively lower compared to soft/moderately hard water.
(4) Ca(ClO)2 exhibits a strong chlorine odor (due to its reaction with water molecules in the air).
Calcium hypochlorite is well-known as the active ingredient in many commercial bleaching agents (such as bleaching powder, chlorinated lime, and chlorinated powder). Its primary application is in disinfecting public swimming pools. It is also used for water disinfection in drinking water. Some other uses of calcium hypochlorite include:
(1) It is useful as an oxidizing agent in organic chemistry.
(2) It is employed to obtain fragmented aldehydes/carboxylic acids by cleaving bonds in glycols and keto acids.
(3) Ca(ClO)2 can also be used in the haloform reaction to produce chloroform.
(4) The compound is utilized for disinfecting wastewater and drinking water due to its high chlorine availability.
(1) Universality and effectiveness across various applications
Choosing calcium hypochlorite formulations over other compounds offers several distinct advantages across different applications. A notable benefit lies in its multifunctionality and efficacy as a disinfectant and bleaching agent. Calcium hypochlorite exhibits broad-spectrum antibacterial activity, effectively neutralizing bacteria, viruses, and algae in water treatment processes. Additionally, its oxidizing properties make it highly effective in bleaching applications, removing stains and improving the appearance of fabrics and surfaces. This versatility allows calcium hypochlorite to be used across a wide range of industries, including municipal water treatment, textile manufacturing, and household cleaning.
(2) Long-term stability and shelf life advantages
Another advantage of calcium hypochlorite formulations is their long-term stability and shelf life, aiding in cost savings and operational convenience. Unlike some alternative compounds that may degrade over time or require special storage conditions, calcium hypochlorite maintains its potency and efficacy over extended periods even when stored in less than ideal environments. This stability ensures a reliable supply of disinfectants and bleaching agents, reducing the need for frequent replenishment and minimizing downtime in industrial processes. Furthermore, the solid form of calcium hypochlorite allows for easy handling and dosing, further enhancing its practicality and applicability across various uses.
The corrosive nature of calcium hypochlorite makes it a hazardous substance that must be carefully stored. It should be kept in a cool, dry environment away from any metals or acids. Contact with any acid will produce highly toxic chlorine gas fumes.
When storing and handling calcium hypochlorite, certain safety guidelines must be followed to ensure the safety of the environment and personnel: (1) During storage, calcium hypochlorite should be kept in a cool, well-ventilated, dry place and should be stored separately from reducing agents, acids, flammable substances, etc. Avoid direct sunlight, and it should be noted that calcium hypochlorite should not be stored in large quantities or for long periods. Additionally, storage containers should be regularly inspected and replaced to ensure the stability and safety of calcium hypochlorite. (2) When handling calcium hypochlorite, it should be done in a well-ventilated environment to avoid inhaling its strong chlorine odor. If using calcium hypochlorite for water and pool disinfection, respiratory masks and protective clothing should be worn. Special protective equipment such as plastic gloves and goggles should be used during handling. In case of splashing, affected areas should be immediately rinsed with plenty of water and disinfected to prevent harm to humans. (3) The handling of calcium hypochlorite should comply with environmental regulations, preferably processed at professional sewage treatment plants, and should not be disposed of or discharged arbitrarily.
Calcium hypochlorite is a cornerstone of disinfection, bleaching, and water treatment across various industries. Its efficacy in neutralizing pathogens, removing stains, and ensuring water safety underscores its indispensable role in maintaining public health and hygiene. Furthermore, the widespread application of calcium hypochlorite, from municipal water treatment plants to textile manufacturing and household cleaning, highlights its versatility and adaptability to diverse needs. Readers are encouraged to further explore the applications of calcium hypochlorite. By harnessing the potential of calcium hypochlorite, both businesses and individuals can enhance the effectiveness and efficiency of their operations while contributing to a cleaner, safer environment. Therefore, readers are urged to take action and consider incorporating calcium hypochlorite into their processes to reap the benefits of its multifunctional disinfection and bleaching properties.
[1] https://byjus.com/chemistry/calcium-hypochlorite/
[2] Hua Wen, Han Pinxin, Shi Wenju, et al. Discussion on the Classification of Transportation Hazard of Calcium Hypochlorite Products and Packaging Technology[J]. Fine and Specialty Chemicals, 2020, 28(05): 23-26. DOI: 10.19482/j.cn11-3237.2020.05.06.
[3] https://pubchem.ncbi.nlm.nih.gov/compound/24504
 |
 |
 |