
Phthalazine is an organic heterocyclic compound, also known as 2,3-diazaphenalene. Phthalazine-1(2H)-one and 2,3-dihydrophthalazine-1,4-dione are two important functional forms of phthalazine. What are the biological activities of phthalazine? Current research indicates that phthalazine exhibits widespread biological activities, such as antitumor, antibacterial, anti-inflammatory, and anti-spasmodic effects. Phthalazine derivatives also possess vasodilatory properties and can be used in the treatment of hypertension. Extensive studies on its chemical properties show that introducing structural multifunctionality can enhance its efficacy in various biological activities, including anti-cancer, anti-seizure, antihypertensive, cardiotonic, anti-diabetic, analgesic, antipsychotic, antimicrobial, antithrombotic, vasodilatory, anti-inflammatory, anti-trypanosomal, antileishmanial, asthma, chronic obstructive pulmonary disease, and more. Drugs containing phthalazine, especially inhibitors of poly(ADP-ribose) polymerase (PARP), phosphodiesterase (PDE), and aldose reductase (AR), exhibit excellent enzyme inhibition activity. Phthalazine is significant for its antitumor activity in leukemia, breast cancer, colon cancer, lung cancer, and kidney cancer. Many pharmaceutical molecules containing phthalazine as a pharmacophore are available on the market and are undergoing clinical development to elucidate the utility of phthalazine as a core molecule.
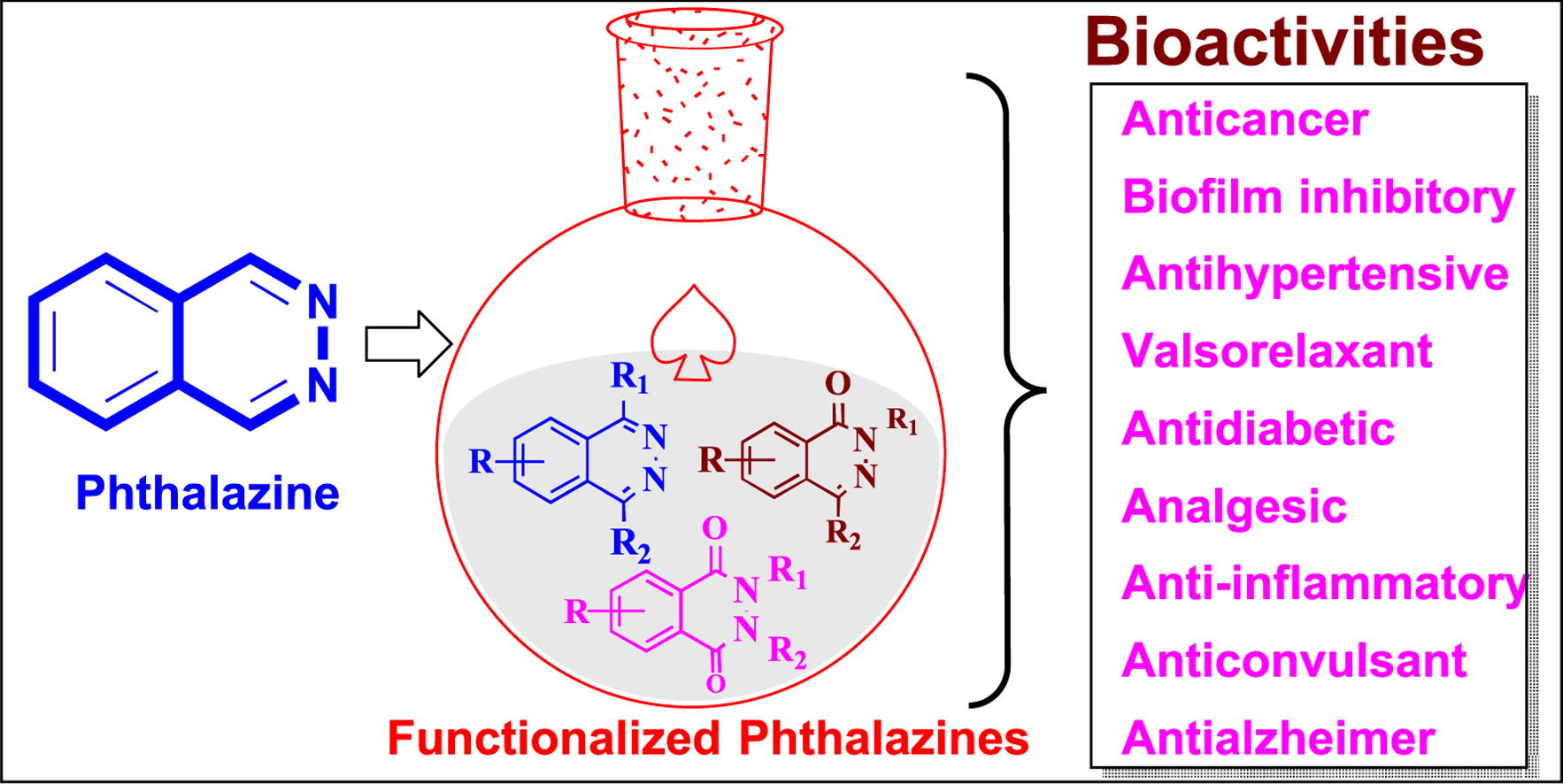
Phthalazine is an important nitrogen-containing heterocyclic compound and serves as a fundamental structural framework for many natural products and drugs synthesized artificially. Phthalazine derivatives are of significant interest due to their extensive biological activities and medicinal value:
In agriculture, phthalazine compounds such as 1-aryl-4-chlorophthalazine have been found to exhibit some herbicidal activity.
Phthalazine can also serve as a reagent in organic chemical reactions. The most reported reaction involving phthalazine is the inverse electron demand Diels–Alder reaction (abbreviated as IED–DA or DAINV reaction):
In the DAINV reaction, phthalazine typically acts as a diene, reacting with an electron-rich dienophile to form naphthalene derivatives after nitrogen elimination (as shown in the figure).
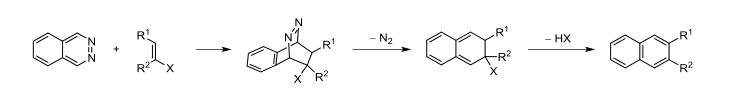
When phthalazine is substituted with electron-withdrawing groups at the 1,4-positions, the DAINV reaction is more likely to occur. However, unsubstituted phthalazine has a higher LUMO orbital energy, making it less reactive with dienophiles, hence the DAINV reaction of phthalazine as a starting material is less frequently reported.
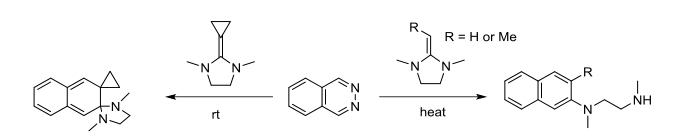
In 1987, Heuschmann and others studied the reaction of phthalazine with highly active dienophiles like 2-alkyl imidazolines. It was found that when the 2-position of imidazoline is a cyclopropyl group, due to increased double bond tension and higher energy, the double bond more readily undergoes cycloaddition, thus completing the reaction at room temperature. However, due to the lack of hydrogen atoms at the α-position, aromaticity cannot be achieved, resulting in the formation of spirocyclic structures. Conversely, when the 2-position of imidazoline is methylene or ethylene, the reaction requires heating, allowing imidazoline to open and further aromaticize to produce naphthalene derivatives.
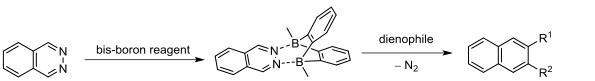
In 2010, Wegner and others cleverly designed a structure to use diborane reagents to catalyze the DAINV reaction of phthalazine. The diborane reagent, acting as an electron-deficient Lewis acid, significantly reduces the electron density of phthalazine, lowering its LUMO orbital energy, as confirmed by computational chemistry. Researchers further used diborane catalysts for the DAINV reaction of phthalazine with electron-rich alkenes such as oxazolines, enamines, or enol ethers, resulting in the formation of naphthalene derivatives with moderate to excellent yields.
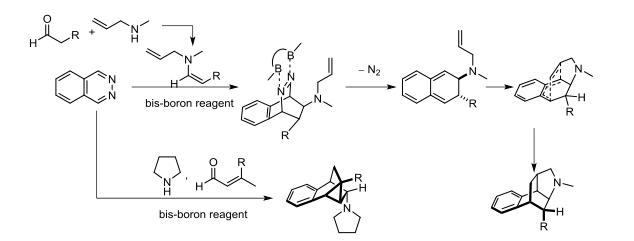
In 2016, Wegner's group again reported a domino reaction based on the diborane/phthalazine system. When using saturated aldehydes and allyl amines as raw materials, the in situ formation of enamines occurs. The enamine double bond undergoes a DAINV reaction with phthalazine, initially forming a 2,3-dihydronaphthalene structure, which then further reacts with the allyl group on the nitrogen atom in a forward D–A reaction to form polycyclic bridge structures. Similar reaction pathways with α, β-unsaturated aldehydes and tetrahydropyrrole yield more complex bridge structures with tricyclic rings.
Phthalazine, as an organic compound with a unique structure, shows broad application prospects in many fields. Its molecular structure imparts excellent electronic properties and coordination abilities, making it a focal point in materials science and drug chemistry. However, challenges in the synthesis and functionalization of phthalazine still exist, such as harsh reaction conditions and low yields. Additionally, the biological activity research of phthalazine derivatives is relatively limited, which restricts its application in drug development. In the future, with continuous improvements in synthesis methods and deeper research into phthalazine properties, it may play a significant role in more fields.
[1]https://go.drugbank.com/categories/DBCAT001325
[2]Luo Mingjian. Study on the addition reaction of phthalazine salts with terminal alkynes[D]. Heilongjiang: Harbin Institute of Technology, 2019.
[3]Zheng Xuanming. Synthesis of phthalazine-based α-aminophosphonates[D]. Heilongjiang: Harbin Institute of Technology, 2018. DOI:10.7666/d. D01587313.
[4]https://en.wikipedia.org/wiki/Phthalazine
[5]Sangshetti J, Pathan S K, Patil R, et al. Synthesis and biological activity of structurally diverse phthalazine derivatives: A systematic review[J]. Bioorganic & Medicinal Chemistry, 2019, 27(18): 3979-3997.
[6]https://www.sciencedirect.com/topics/medicine-and-dentistry/phthalazine
[7]https://www.sciencedirect.com/science/article/abs/pii/S0968089619310193
[8]Zhao Meimei, Tong Hongjuan, Du Mo. Research progress on the synthesis and activity of phthalazine derivatives [J]. Fine and Specialty Chemicals, 2022, 30(4): 15-19. DOI: 10.19482/j.cn11-3237.2022. 04.03.
 |
 |
 |